TN State Board 11th Chemistry Important Questions Chapter 11 Fundamentals of Organic Chemistry
Question 1.
Name the organic compounds found in living things?
Answer:
DNA — Deoxyribonucleic acid, glycolipids, glycogen, lipoproteins, phospolipids etc…
Question 2.
Write the state of hybridization of all atoms in CH2 = C = CH2.
Answer:
The compound is known as alkene.
3 2 1
CH2 = C = CH2
All the three carbon atoms are joined by double bonds. Hence all of them are sp2 hybridised.
![]()
Question 3.
Why does carbon undergo hybridisation prior to bond formation?
Answer:
Carbon forms four covalent bonds which of equal length and a bond angle of 109°28′ in alkanes, 120° in alkenes and 180° in alkynes. Overlapping of V and / or ‘p’ orbitals does not explain the bond length and bond angles. Hybridisation involves the formation of hybrid orbitals of equal energy and oriented in space in specific directions. It explains the experimentally observed bond angles and bond length in organic compounds.
Question 4.
What is the type of hybridisation of each carbon in the following compounds?
(i) CH3Cl
(ii) (CH3)2CO,
(iii) CH3CN,
(iv) HCONH2
(v) CH3CH = CHCN
Write partly condensed formula of the above compounds and indicate the type of hybridisation in each compound.
Answer:
(i) 
(ii) 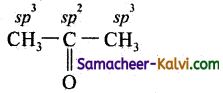
(iii) 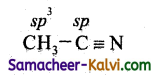
(iv) 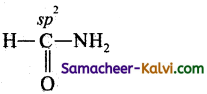
(v) 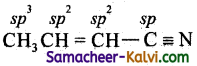
![]()
Question 5.
Expand each of the following condensed formulae into their complete structural formulae,
(i) CH3CH2COCH2CH3
Answer:
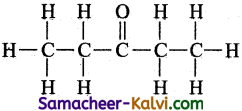
(ii) CH3CH==CH(CH2)3 CH3
Answer:
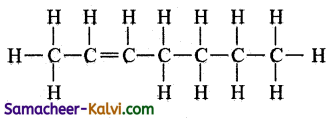
Question 6.
For each of the following write their condensed formula and also their bond line formula.
(i) HO CH2 CH2 CH2 CH(CH3) CH(CH3) CH3
(ii) NaC ≡ CH — C = N
Answer:
Condensed formulae:
(i) HO.(CH2)3 CH(CH3)CH(CH3)2
(ii) HO CH(CN)2
Bond line formulae:
(i) 
(ii) 
![]()
Question 7.
What are alicyclic compounds? Give two examples with their names.
Answer:
Carbocyelic compounds which resemble aliphatic compounds in most of their properties are called alicyclic compounds.
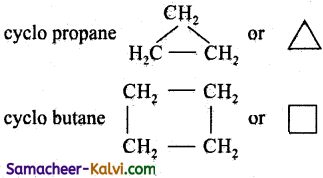
Question 8.
What are homocyelic and heterocyclic compounds? Give an example of each type along with its name.
Answer:
Homocyelic compounds contains rings which are made up of only one kind of atoms i.e., carbon atoms.
eg: cyclo hexane or benzene.
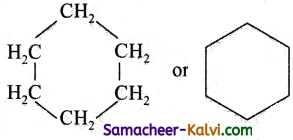
Heterocyclic compounds contain one or more hetero atoms (i.e., atoms other than carbon and hydrogen, eg: Oxygen, Sulphur, Nitrogen) in the ring.
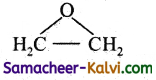
Question 9.
Write the functional groups of the following.
(i) alcohol
Answer:
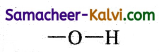
(ii) mono carboxylic
Answer:
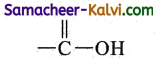
(iii) aldehyde
Answer:
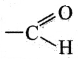
(iv) ketone
Answer:
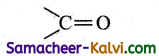
(v) ester
Answer:
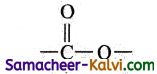
![]()
Question 10.
Indicate primary prefix, root word, primary suffix and secondary suffix in the following compounds and write their IUPAC name.
Answer:
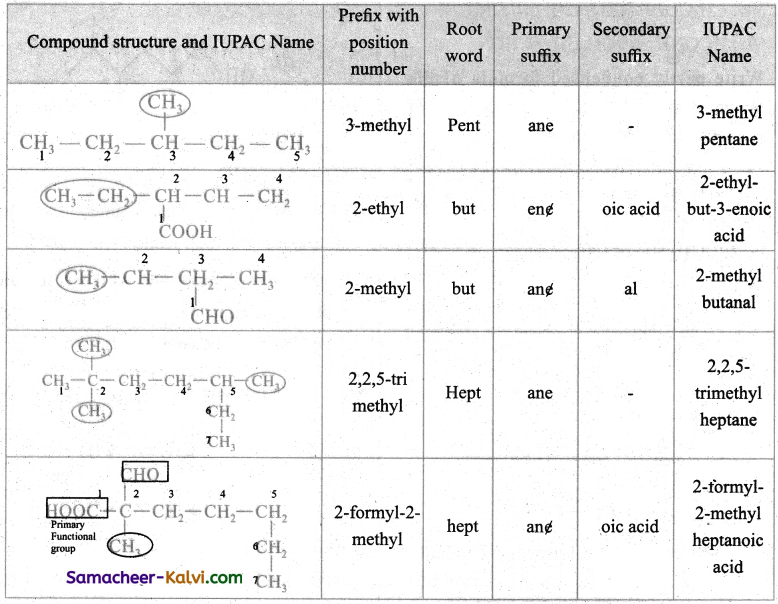
Question 11.
Explain the terms (i) Root word (ii) Prefix and (Hi) Suffix’in writing the name of an organic compound.
Answer:
The IUPAC name of an organic compound consists of three parts.
prefix + root word + suffix .
(i) Root word denotes the number of carbon atoms in the longest continuous chain in molecules
(ii) Prefix denotes the group(s) attached to the main chain which is placed before the root.
(iii) Suffix denotes the functional group and is placed after the root word.
![]()
Question 12.
Write the IUPAC names for the following compounds:
(i) 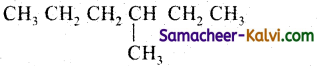
Answer:
Rule 1:
Select the largest continuous chain of carbon atoms in the molecule. This is called the parent chain while all other carbon atom which are not included in the parent chain are regarded as prefixes i.e., side chain or substituents. The largest chain contains 6 carbon atoms and it is named as a derivative of hexane.
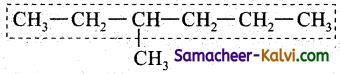
Rule 2:
Numbering of the carbon atom containing the substituent.
The numbering of the carbon atom containing the substituent is done in sucha way that the carbon atom containing the substituent should bear a lower number.
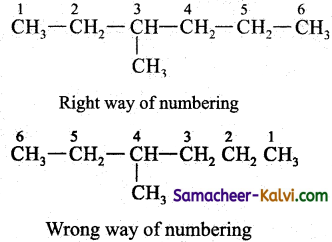
Now the primary prefix is 3-methyl. Root word is hex; primary suffix is ‘ane’; IUPAC name 3-methyl hexane.
(ii) 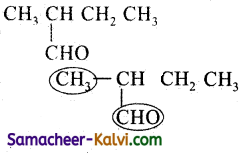
Answer:
Rule 1:
If the organic compound contains two or more functional groups, one of the functional groups is selected as principal functional group while all the remaining functional groups are heated as substituents. The principal functional group in this compound is aldehyde group.
Rule 2:
When a chain terminating functional group such as —CHO,—COOH,—COOR, – CONH2,—COCl,—C ≡ N, etc is present, it is always given number 1.
Hence the numbering of the above compound is
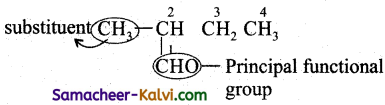
Prefix : 2-methyl; Root word is but; Primary prefix – an£; Secondary prefix = al; IUPAC name: 2-methyl butanal.
(iii) 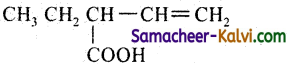
Answer:
The principal functional group is —COOH (carboxylic acid group) and it should be always numbered as 1 and followed by double bond, triple bond and substituents in that order. Hence naming is done in the order and group, double bond and substituent.
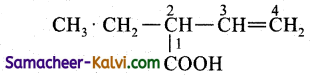
Prefix: 2-ethyl; Root word is but; primary prefix = 3 – en; secondary suffix is oic acid. Here the IUPAC name is 2-ethyl-3-enoic acid.
(iv) 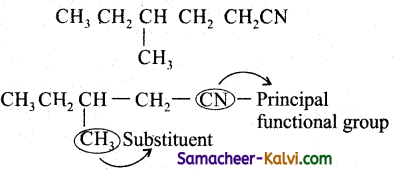
Answer:
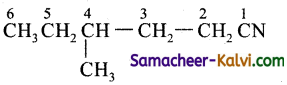
Rule as given in (iii)
Primary prefix = 4- methyl;
Root word = hex;
Primary suffix = an;
Secondary suffix – nitrile.
IUPAC name = 4-methyl hexane nitrile.
(v) 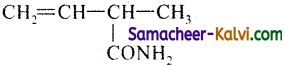
Answer:
Rule as given in (iii)
Primary functional group = CONH2
Substitution CH2 = CH
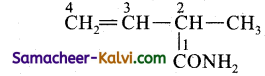
Primary prefix = 2-methyl;
Root word = but;
Primary suffix = 3- en;
Secondary suffix – amide.
IUPAC name = 2-methyl but-3-enamide.
![]()
Question 13.
Write the condensed structural formula for the following compounds. Identify the prefix with position number, root word, primary prefix, secondary suffix and write them in the same order.
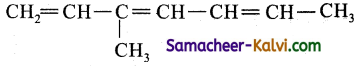
(i) 3-methyl hepta-1, 3, 5 triene;
Answer:
The root word is ‘hept’. This means the carbon chain contains 7 carbon atoms.
C — C — C — C — C — C — C
Number them from either end
![]()
The primary prefix is 3 methyl i.e., methyl group is present as a substituent in the 3rd carbon atom. .
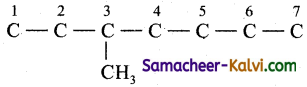
The primary suffix is 1, 3, 5 triene, i.e., similarly the carbon atom 1, 3, 5 contain double bonds.
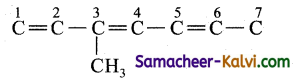
Now complete the valencies of carbon by single bond with hydrogen.
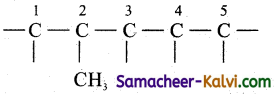
(ii) 2-methyl pentan-1-ol:
Answer:
The root word is ‘pent’. This means the carbon chain contains 5 carbon atoms,
—C—C—C—C—C
Number the carbon atom from any end. The primary prefix with position number is 2-methyl. Hence put the methyl group on a substituent in 2nd carbon atom.
The primary suffix is ane and the secondary suffix is ‘ol’.
Hence put the ‘OH’ group in the second carbon atom and fill up the necessary hydrogen atom.
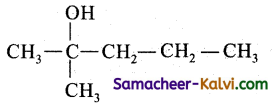
(iii) Propanoic acid:
Answer:
The root word is ‘prop’ i.e., the carbon chain contains 3 carbon atoms.
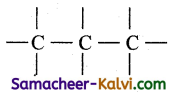
Since the primary suffix is ang and secondary suffix is ‘oic acid’, number the carbon atom from either end and place 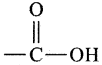 group at the terminal carbon atom
group at the terminal carbon atom
Fill up the hydrogen atom. Hence the structure is CH3CH2COOH.
![]()
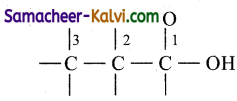
(iv) Pentan-3-one:
Answer:
Root word is pent. Draw the carbon chain containing 5 carbon atoms.
Since secondary suffix is 3-one, put C = O in the third carbon atom.
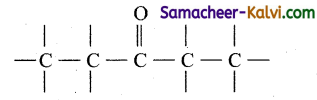
Fill the hydrogen atoms.
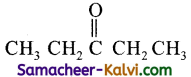
(v) 2-cyclopentyl propanal:
Answer:
Primary prefix = 2 cyclo pentyl;
Root word = prop;
Primary suffix = an£;
Secondary suffix = al.
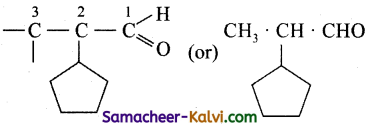
![]()
Question 14.
Draw the condensed structure and bond line structure of the following;
(i) 1 – 3 – butadiene:
Answer:
Condensed structure:
CH2 = CH — CH = CH2
Bond line structure:

(ii) tert-butyl alcohol:
Answer:
Condensed structure formula:
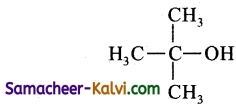
Bond line structure:
![]()
(iii) 4-ethyl-2-4-dimethyl hept-1-ene:
Answer:
Condensed structure formula:
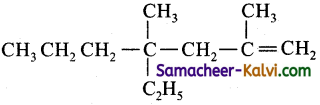
Bond line structure:
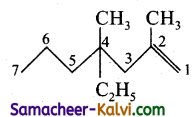
(iv) 3 methyl but – 1 – yne:
Answer:
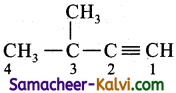
Bond line structure:
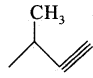
![]()
(v) 3 chloro – 1 – phenyl prop – 1 – ene
Answer:
Condensed structure formula:
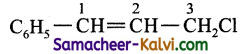
Bond line structure:
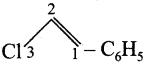
(vi) 1 – bromo but 1- 1 – ene:
Answer:
Condensed structure formula:
![]()
Bond line structure:
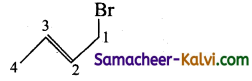
(vii) 2 chloro-3-ethyl penta 1,4 diene:
Answer:
Condensed structure formula:
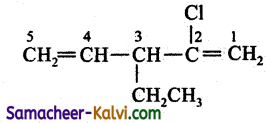
Bond line structure: Cl
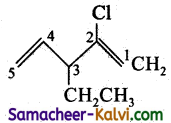
![]()
Question 15.
What type of isomerism is exhibited by the following compounds? Write the structure of isomers,
(i) Pentane
(ii) but-1-ene
(iii) butan-1 -ol
(iv) Pentanal
(v) pent-1 -yne
Answer:
All of them exhibit chain isomerism i.e., chain isomer differ in the arrangement of carbon atom in the chain.
(i) CH3 CH2 CH2 CH2 CH3; n – pentane
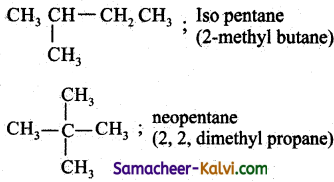
(ii) CH3 CH2 CH = CH3;
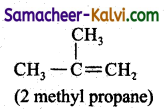
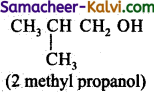
(iii) CH3 CH2 CH2 CH2 OH ; CH3 CH- CH2 OH
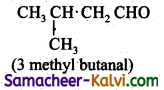
(iv) CH3 CH2 CH2 CH2 CHO ; pentanal
(v) HC ≡ C CH2 CH2 CH3; pent-1-yne
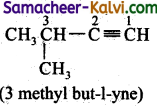
Question 16.
Select the pair of chain isomers among the following:
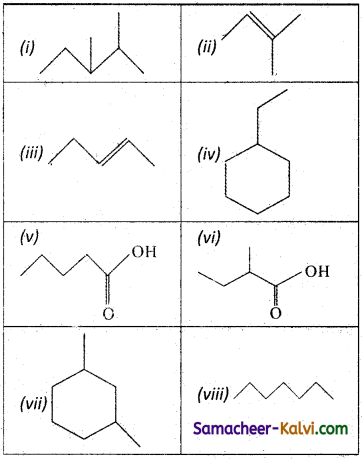
Answer:
(ii) and (iii), (iv) and (vi); (v) and (vi); (i) and (viii).
![]()
Question 17.
Write down all the structural isomers of C6H14. Indicate the chain and position isomers among them.
Answer:
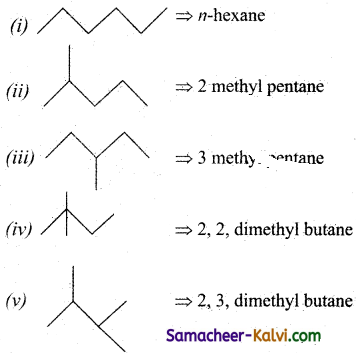
Chain isomers: (i), (ii), (iii), (iv), (v)
Position isomers:
(a) (ii) and (iii),
(b) (iv) and(v).
Question 18.
Explain the term position isomerism with an example.
Answer:
It arises due to the difference in positions occupied by the substitu ents or due to different positions of double bond or triple bond in the case of alkenes and alkynes respectively.
eg: 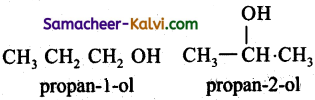
![]()
Question 19.
Write the structures of the isomers having the molecular formulae.
(i) C4H8
Answer:
CH3CH2CH = CH2;
But – 1 – ene
CH3 CH = CHCH3
But – 2 – ene
(ii) C4H6
Answer:
CH3CH2C ≡ CH ;
But – 1 – yne
CH3C ≡ C CH3
But – 2 – yne
(iii) C3H7Cl
Answer:
CH3CH2CH2Cl;
1 – Chloropropane
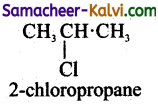
(iv) C6H4Cl2
Answer:
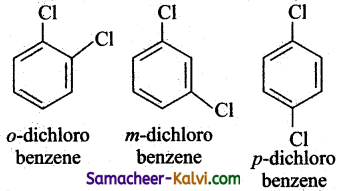
(iii)C8H10
Answer:
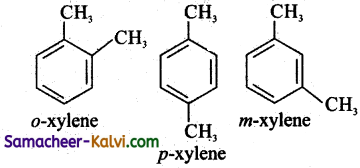
![]()
Question 20.
Explain the term stereoisomerism.
Answer:
(i) Stereoisomerism arises due to the difference in spatial arrangement of atoms or groups in the same molecule.
(ii) There are two types of stereo isomerism. They are geometrical and optical isomerism.
(iii) Restricted rotation of C = C in alkenes results in the formation of two isomers where in ‘cis’ isomer, two identical groups are present on the same side of C = C double bond. In ‘trans’ isomer the two identical groups lie opposite to each other across C = C double bond.
eg: 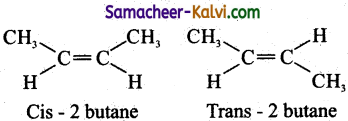
(iv) These isomers have different physical properties.
(v) Optical isomerism arises due to molecular asymmetry. Compounds which possess non-super impossible object-mirror image relationship exhibit optical isomerism.
(vi) Dextro and laevo isomers (also known as enantiomers) rotate the plane of the plane polarised light to the right and left respectively.
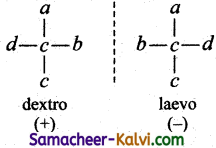
![]()
Question 21.
Trans isomer is more stable than cis isomer. Explain why?
Answer:
The trans isomer is more stable than the corresponding cis isomers. This is because in the cis isomer, the bulky groups are on the same side of the double bond. The steric repulsion of the groups makes the cis isomers less stable than the trans isomers in which bulky groups are on the opposite side.
Question 22.
Explain the geometrical isomerism exhibited oximes.
Answer:
Restricted rotation around C = N (oximes) gives rise to gieometrical isomerism in oximes. Here ‘syn’ and ‘anti’ are used instead of cis and trans respectively. In the syn isomer the H atom of a doubly bonded carbon and – OH group of doubly bonded nitrogen lie on the same side of the double bond, while in the anti isomer, they lie on the opposite side of the double bond.
For eg: 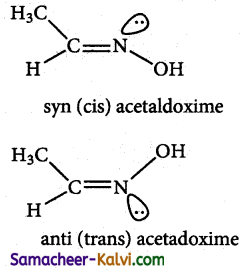
Question 23.
What are enantiomers? Give an example.
Answer:
Enantiomers are optically active substances, which possess a chiral carbon atom and possess non-super impossible object-mirror image relationship.
eg: 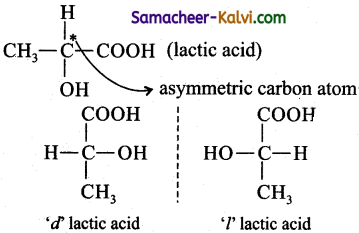
![]()
Question 24.
Explain the term qualitative analysis of an organic compound.
Answer:
The elements present in the organic compound mainly carbon and hydrogen and other elements like halogen, sulphur, phosphorous and nitrogen. The presence of these elements are detected by performing chemical tests. This process is known as qualitative analysis, i.e., analyzing the compound and identifying them by their chemical properties.
Question 25.
Mention the principle involved in the detection of carbon and hydrogen in an organic compound.
Answer:
The carbon present in an organic is oxidised to carbon-dioxide and the hydrogen is oxidised to water. By cupric oxide the reactions involved are
C + 2 CuO → CO2 + 2 Cu
2 H + CuO → H2O + Cu
The carbon-dioxide is tested by passing the gas through lime water, which is turned milky. This indicates the presence of carbon.
The water formed in the above reaction, when sprinkled into white anhydrous copper sulphate, it turns blue. This indicates the presence of hydrogen in an organic compound.
![]()
Question 26.
Describe the preparation of sodium fusion extract, to detect presence of nitrogen in an organic compound.
Answer:
A freshly dried piece of sodium metal is taken in with the organic compound in fusion tube and heated strongly till it become red hot. The red hot fusion tube is suddenly dipped into cold water taken in a china dish. The contents are boiled for 10 minutes and then filtered. The filtrate is known as sodium fusion extract and used for detection of nitrogen, sulphur, phosphorous and halogen. The extract is also known as Lassaigne extract.
Question 27.
Why is it necessary to heat the organic compound with sodium metal, while preparing sodium fusion extract?
Answer:
The organic compounds being polar are insoluble in water. By heating the organic compound with sodium, the elements form soluble ionic compounds with sodium. This makes analysis in aqueous solution easier VIZ, NaCN, Na2S, NaCl, etc…
Question 28.
How will you detect nitrogen present in an organic compound?
Answer:
To 2 ml of sodium fusion extract, freshly prepared ferrous sulphate is added. A green precipitate of ferrous hydroxide appears. The mixture is boiled for 2 minutes, cooled and acidified with dilute sulphuric acid (H2SO4 dissolves ferrous hydroxide). Appearance of a green or prussian blue confirms the presence of nitrogen in the compound. (Alternatively, FeCl3 and dil HCl may be added).
![]()
Question 29.
Brief explain with equations the chemistry of Lassaigne test for detecting nitrogen in an organic compound.
Answer:
Lassaigne’s filtrate solution consists of sodium cyanide and sodium hydroxide
Na + ((C + N) Trans org. compound) → NaCN (Sodium cyanide)
2 Na + 2H2O → 2NaOH + H2
When the filtrate is boiled with ferrous sulphate solution, the following reactions take place. FeSO4 + 2NaOH
FeSO4 + 2 NaOH → Fe(OH)2 (Ferrous hydroxide (green)) + Na2SO4
Fe(OH)2 + 6NaCN → Na4[Fe(CN)6] (sodium ferro cyanide) + 2NaOH
At the same time, ferrous sulphate is converted to ferric sulphate. This reacts with sodium ferro cyanide to form ferric ferro Cyanide which gives green or prussian blue colour ro precipitate.
3 Na4[Fe(CN)6] + 2 Fe2(SO4)3 → Fe4[Fe(CN)6] (ferric ferro cyanide (Prussian blue)) + 6 Na2SO4
Question 30.
What happens when both nitrogen and sulphur in the organic compound. How will they be detected by Lassaigne’s test?
Answer:
In case if both N & S are present, a blood red color is obtained due to the following reactions.
Na + C + N + S ![]() NaCNS (sodium sulphocyanide)
NaCNS (sodium sulphocyanide)
3 NaCNS + FeCl3 → Fe(CNS)3 (ferric sulphocyanide (Blood red color)) + 3 NaCl
![]()
Question 31.
If sulphur Or nitrogen is present in an organic compound along with halogens, how will you detect the halogens.
Answer:
If sulphur is present along with nitrogen and halogens, the sodium fusion extract will contain Na2S (Sodium sulphide) along with NaCN. Both NaOH and Na2S, will react with silver nitrate (added to detect halogen) and form a white precipitate of AgCN and a black precipitate of Ag2S. Thus, the halogens in the compound cannot be detected.
Hence, to remove NaCN and Na2S, Sodium fusion extract is boiled with concentrated nitric acid which decomposes NaCN and Na2S.
NaCN + HNO3 → NaNO2 + HCN↑
Na2S + 2 HNO3 → 2NaNO3 + H2S↑
The solution free from NaCN and Na2S is cooled, and silver nitrate is added after acidifying with dilute nitric acid. A white precipitate (AgCl), soluble in ammonia indicates presence of chlorine. A pale yellow precipitate, sparingly soluble in ammonia indicates the presence of bromine. A yellow precipitate insoluble in ammonia indicates the presence of iodine in the organic compound.
Question 32.
Give the reaction involved in the detection of halogen in an organic compound.
Answer:
Na + X (from organic compound) ![]() Na X (where x = Cl, Br, I, F)
Na X (where x = Cl, Br, I, F)
NaX + AgNO3 → AgX + NaNO3
![]()
Question 33.
Explain how sulphur is detected in an organic cojnpound by qualitative analysis.
Answer:
| Experiment | Observation and inference |
| Sodium fusion extract + freshly prepared sodium nitro prusside solution. | A deep violet colour indicates the presence of sulphur. |
| Sodium fusion extract + acetic acid + lead acetate solution. | A black precipitate indicates the presence of sulphur. |
| Organic substance + KNO3 + Na2CO3 6186 |
A white precipitate of BaSO4 indicates the presence of sulphur. |
Question 34.
Explain how the chlorine, bromine and iodine present in an organic compound is detected.
Answer:

![]()
Question 35.
Explain how will you detect phosphorous in an organic compound.
Answer:
The organic compound is fused with a mixture of sodium carbonate and potassium nitrate (fusion mixture). The phosphorus present in the compound is converted to sodium phosphate. The fused product is extracted with water and the aqueous solution obtained is boiled with concentrated nitric acid and ammonium molybdate. A yellow precipitate or yellow colouration indicates the presence of phosphorus. The yellow precipitate is due to the formation of ammonium phosphomolybdate (NH4)3 PO4.12MoO3.
Question 36.
Explain the principle involved in the estimation of carbon and hydrogen in an organic compound.
Answer:
Both carbon and hydrogen are estimated by the same method. A known weight of the organic substance is burnt in excess of oxygen and the carbon and hydrogen present in it are oxidized to carbon-dioxide and water, respectively.
Cx Hy + O2 ![]() xCO2 + y H2O
xCO2 + y H2O
The weight of carbon-dioxide and water thus formed are determined and the amount of carbon and hydrogen in the organic substance is calculated.
The percentage of carbon and hydrogen are calculated by the following formulae:
Percentage of Carbon = \(\frac{12}{44}\) × (Weight of CO2 formed / Weight of organic substance) × 100
Percentage of carbon = \(\frac{2}{8} \times \frac{\text { Weight of } \mathrm{H}_{2} \mathrm{O} \text { formed }}{\text { Weight of the organic compound }} \times 100\)
![]()
Question 37.
Explain the principle involved in the estimation of sulphur in an organic compound (by carius method).
Answer:
A known mass of the organic substance is heated strongly with fuming HNO3. C & H get oxidised to CO2 & H2O while sulphur is j oxidised to sulphuric acid as per the following j reaction.
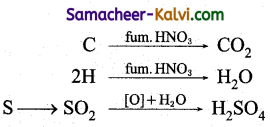
The resulting solution is treated with excess of BaCl2 solution H2SO4 present in the solution in quantitatively converted into BaSO4, from the mass of BaSO4, the mass of sulphur 1 and hence the percentage of sulphur in the compound Can be calculated.
The percentage of sulphur is calculated by using the formula
Percentage of Sulphur = \(\frac{32}{233} \times \frac{\text { Weight of } \mathrm{BaSO}_{4} \text { formed }}{\text { Weight of the organic compound }} \times 100\)
![]()
Question 38.
How will you estimate the halogens present in an organic compound?
Answer:
A known mass of the organic compound is heated with fuming HNO3 and AgNO3. C, H & S get oxidised to CO2, H2O & SO2 and halogen combines with AgNO3 to form acipitate of silver halide.
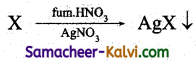
The ppt of AgX is filtered, washed, dried and weighed. From the mass of AgX and the mass
of the organic compound taken, percentage of halogens are calculated.

Question 39.
Explain how phosphorus in an organic compound is estimated?
Answer:
A known mass of the organic compound is heated with fuming HNO3 in a sealed tube. The carbon and hydrogen are oxidized to CO2 and H2O respectively. While phosphorus
present in the organic compound is oxidised to phosphoric acid (H3PO4). This is precipitated as ammonium phospho molybdate by heating with Conc.HNO3 and then adding ammonium molybdate. From the weight of ammonium phospho molybdate, the percentage of phosphorus can be determined.
In an alternative method, the phosphoric acid is precipitated as magnesium-ammonium
phosphate by adding magnesia mixture (a mixture containing MgCl2, NH4Cl and ammonia) This ppt is washed, dried and ignited to get magnesium pyrophosphate which is washed, dried a weighed.

![]()
Question 40.
Explain the princ iple involved for the preparation of nitrogen man organic compound by Dumas method.
Answer:
A known mass of the organic compound j is heated with excess of cupric oxide in an atmosphere of CO2. Carbon, hydrogen and sulphur if present are oxidised to CO2, H2O and SO2 respectively, while nitrogen gas is set free. Any oxide of nitrogen that may be formed is reduced to free N2 by passing over heated copper.
If the organic compound assigned the molecular formula Cx Hy Nz, the combustion ) equation is
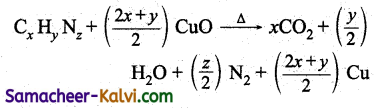
The nitrogen formed is collected over KOH and from the volume of N2 formed, the percentage can be calculated.
Percentage of N2 = \(\frac{28}{22.4} \times \frac{\text { Volume of } \mathrm{N}_{2} \text { formed at NTP }}{\text { Weight of the organic compound }} \times 100\)
Note:
The volume of N2 formed at the experimental temperature and pressure should by using the formula
\(\frac{P_{1} V_{1}}{T_{1}}=\frac{P_{2} V_{2}}{T_{2}}\)
Question 41.
Explain the principle involved in Kjeldahls method of estimation of nitrogen.
Answer:
(i) Kjeldahls method is based on the fact that when an organic compound containing nitrogen is heated With cone. H2SO4, the nitrogen in it is quantitatively converted to ammonium sulphate. The resultant liquid is then treated with excess of alkali \ and then liberated ammonia gas absorbed in excess of standard acid. The amount of ammonia (and hence nitrogen) is determined by finding the amount of acid neutralized by back titration with same standard alkali.
From the volume and the normality of the acid used to react with NH3, the percentage of nitrogen present in the organic compound can be determined by using the formula,
Percentage of Nitrogen = \(\frac{14 \times \mathrm{N} \times \mathrm{V}}{1000 \times \mathrm{w}} \times 100\)
Where N is the strength of H2SO4 used to neutralize NH3 and V in the volume of H2SO4 required for the complete neutralisation of evolved ammonia and ‘ w’ is the weight of the organic compound.
![]()
Question 42.
0.92 g of an organic compound containing carbon, hydrogen and oxygen was analysed by combustion method. The increase in mass of the U tube and the potash bulbs at the end of the experiment was found to>be 1.08g and 1.76g respectively. Determine the percentage composition of carbon, hydrogen and oxygen in the compound.
Answer:
Increase in mass of ‘U’ tube = 1.08 g
i.e., mass of water formed = 1.08 g
Since, 18 g of H2O contain = 2 g of hydrogen
1.08 g of H2O contain = \(\frac{2}{18}\) × 1.08 g of hydrogen
percentage of hydrogen = \(\frac{2}{18}\) × \(\frac{1.08}{0.92}\) × 100 = 13.04
Increase in mass of potash bulbs = 1.76 g
Mass of carbon-dioxide formed = 1.76 g
Since, 44 g of CO2 = 12 g of Carbon
1.76 g of CO2 = \(\frac{12}{44}\) × 1.76 g of carbon
percentage of carbon = \(\frac{12}{44}\) × \(\frac{1.08}{0.92}\) × 100 = 52.17
percentage of oxygen = 100 – (%C + % H)
= 100 – (52.17 + 13.04) = 34.79
Question 43.
On complete combustion of 0.246 g of an organic compound gave 0.198 g of carbondioxide and 0.1014 g of water. Determine the percentage combustion of carbon and hydrogen in the compound.
Answer:
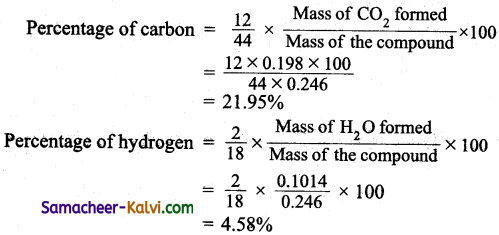
![]()
Question 44.
An organic compound was analysed by Duma’s method. The 0.45 g of the organic compound, gave 48.6 ml of nitrogen at 27°C and 756 mm pressure. Calculate the percentage of nitrogen.
Answer:
V1 = 48.6 mL
V2 = ?
P1 = 756 mm
P2 = 760 mm
T1 = 27 + 273 = 300 K
T2 = 273 K
Applying the formula,
Volume of N2 at NTP (V2) = \(\frac{\mathrm{P}_{1} \mathrm{~V}_{1}}{\mathrm{~T}_{1}} \times \frac{\mathrm{T}_{2}}{\mathrm{P}_{2}}=\frac{756 \times 48.6 \times 273}{300 \times 760}\) = 43.99 mL
Mass of organic compound = 0.45 g
percentage of notrogen in the compound = \(\frac{28}{22400} \times \frac{\text { Vol. of } \mathrm{N}_{2} \text { at } \mathrm{NTP}}{\text { Mass of the compound }} \times 100\)
= \(\frac{28}{22400} \times \frac{43.99}{0.45} \times 100\) = 12.22 %
Question 45.
On complete combustion of 0.246 g of an organic compound gave 0.198 g of carbondioxide and 0.1014 g of water. Determine the percentage combustion of carbon and hydrogen in the compound. (Vapour pressure of water aqueous tension at 300 K is 15 mm)
Answer:
Pressure of the gas (N2) = 715 – 15 = 700 mm
Converting the volume of N2 measured at 300 K and 715 mm pressure to the volume corresponding to NTP.
\(\frac{\mathrm{P}_{1} \mathrm{~V}_{1}}{\mathrm{~T}_{1}}=\frac{\mathrm{P}_{2} \mathrm{~V}_{2}}{\mathrm{~T}_{2}}\)
P1 = 700 mm
P2 = 760 mm
V1 = 50 mL
V2 = ?
T1 = 300 K
T2 = 273 K
V2 = \(\frac{700 \times 50}{300} \times \frac{273}{760}\) = 41.9 mL
Weight of the organic compound = 0.30 g
22400 mL of N2 at NTP = 28 g
41.9 mL of N2 at NTP = \(\frac{28}{22400}\) × 41.9 g
0.30 g of the organic compound contain = \(\frac{28}{22400}\) × 41.9 g of N2
100 g of the organic compound will contain 28 = \(\frac{28}{22400} \times \frac{41.9}{0.30} \times 100\) = 17.46 %
![]()
Question 46.
0.2 g of an organic compound was analysed by Kjeldahls method. Ammonia evolved was absorbed in 60 mL of N/5 H2SO4 unused acid required 40 mL of N/10 NaOH for complete neutralisation. Find the percentage of nitrogen in the compound.
Answer:
Mass of organic compound = 0.2 g
Unused acid required = 40 mL \(\frac{\mathrm{N}}{10}\) NaOH
i.e., 40 mL. \(\frac{\mathrm{N}}{10}\) NaOH = 40 mL \(\frac{\mathrm{N}}{10}\) H2SO4
i.e., 40 mL \(\frac{\mathrm{N}}{10}\) H2SO4 = 20 mL of \(\frac{\mathrm{N}}{5}\) H2SO4
Acid used in the absorption of ammonia = (60 – 20) \(\frac{\mathrm{N}}{5}\) H2SO4
= 40 mL of \(\frac{\mathrm{N}}{5}\) H2SO4
Percentage of N2 = \(\begin{gathered}
1.4 \times \text { Normality of the acid used } \\
\quad \times \text { Volume of the acid used } \\
\hline \text { Weight of Substance }
\end{gathered}\)
= \(\frac{1.4 \times \frac{1}{5} \times 40}{0.2}=\frac{1.4 \times 40}{0.2 \times 5}\) = 56 %
Question 47.
During estimation of nitrogen present in an organic compound by Kjeldahls method, the ammonia evolved from (15g of the organic compound, neutralised 10 mL of 1M H2S04. Find the percentage of nitrogen in the cornpound.
Answer:
10 mL of 1M H2SO4 10 mL of 2N H2SO4.
Volume of acid required to react with NH3 = 10 mL .
Normality of the acid used = 2 N
Weight of the compound = 0.58
Percentage of N2 = \(\begin{gathered}
1.4 \times \text { Normality of the acid used } \\
\times \text { Volume of the acid used } \\
\hline \text { Wt. of substance }
\end{gathered}\)
= \(\frac{1.4 \times 2 \times 10}{0.5}\) = 56 %
![]()
Question 48.
1.216 of an organic compound was reacted under Kjeldahls method and the ammonia evolved was absorbed in 100 mL of N H2SO4. The remaining acid solution was made upto 50 mL by the addition 0f water. 20 mL ot the acid solution required 32 mL of \(\frac{\mathrm{N}}{10}\) caustic soda solution br complete neutralisation. Calculate the percentage composition of nitrogen in the organic compound.
Answer:
20 mL of diluted unreacted acid solution required = 32 mL of NaOH
500 mL of dilute unreacted acid solution required= \(\frac{32}{50}\) × 50 mL of \(\frac{\mathrm{N}}{10}\) NaOH solution
= 80 mL of \(\frac{\mathrm{N}}{10}\) NaOH solution
i.e., 80 mL \(\frac{\mathrm{N}}{10}\) NaOH = 80 mL of \(\frac{\mathrm{N}}{10}\) H2SO4
Acid used for neutralisation of NH3 = (100 – 80) mL of N H2SO4
= 20 mL of N H2SO4
Percentage of nitrogen = \(\frac{1.4 \times \mathrm{N}(\text { acid used }) \times \mathrm{V} \text { (acid used) }}{\text { Weight of organic compound }}\)
= \(\frac{1.4 \times 1 \times 20}{1.216}\) = 23.03 %
Question 49.
0.5264 g of silver bromide is obtained from 0.5124 g of m organic compound. Calculate the percentage of bromine in the compound.
Answer:
mass of the compound (W) = 0.5124 g
mass of silver bromide (w) = 0.524 g
1 mole of AgBr = 1 mole of Br
188 g of AgBr = 80 g of Bromine
0.5264 g of AgBr = \(\frac{80}{188}\) × 0.5264 g of Br
0.5124 g of organic compound = \(\frac{80}{188}\) × 0.5264 g of Br
100 g of the organic compound = \(\frac{80}{188}\) × \(\frac{80}{188}\) ×100 g = 43.71 %
![]()
Question 50.
0.156g of an organic compound on heating with fuming HNO3 and AgNO3 gives 0.235g of silver iodide. Calculate the percentage of iodine in the compound.
Answer:
Mass of organic compound = 0.156 g
Mass of AgI formed = 0.235 g
1 mol of AgI = 1 mol of I
235 g of AgI = 127 g of I
0.235 g AgI will contain = \(\frac{127}{235}\) × 0.235 g of iodine
0.156 g of the compound contain = \(\frac{127}{235}\) × 0.235 g of iodine
100 g of the compound contain = \(\frac{127}{235}\) × \(\frac{0.235}{0.156}\) × 100 = 81.41 %
Question 51.
0.1170 g of an organic compound on heating with conc. HNO3 and AgNO3 in a carius furnace gave 0.42g of silver chloride. Find the percentage of chlorine in the organic compound.
Answer:
Mass of organic compound = 0.1170 g
Mass of silver chloride formed = 0.42 g
1 mol of AgCl = 1 mol of Cl
143.5 g of AgCl = 35.5 g of chlorine
0.42 g of AgCl = \(\frac{35.5}{143.5}\) × 0.42 of g chlorine
0. 1170 g of organic compound contain = \(\frac{35.5}{143.5}\) × \(\frac{0.42}{0.1170}\) g of chlorine
100 g of the organic compound will contain = \(\frac{35.5}{143.5}\) × \(\frac{0.42}{0.1170}\) × 100 = 88.80 %
![]()
Question 52.
In sulphur estimation, 0.157 g of an organic compound gave 0.4813 g of barium sulphate. What is the percentage of sulphur in the compound?
Answer:
Weight of organic compound = 0.157 g
Weight of barium sulphate formed = 0.4813 g
BaSO4 = S
1 mol of BaSO4 = 1 mol of sulphur
233 g of BaSO4 = 32 g of sulphur
0.4813 g of BaSO4 = \(\frac{32}{233}\) × 0.4813 g of sulphur
0.157 g of the compound contains = \(\frac{32}{233}\) × 0.4813 g of sulphur
100 g of the compound contains = \(\frac{32}{233}\) × \(\frac{0.4813}{0.157}\) × 100 = 42.10%
Question 53.
On heating 0.32 g of an organic compound with concentrated nitric acid and barium chloride, 0.932 g of barium sulphate was obtained. Calculate the percentage of sulphur in the compound.
Answer:
Mass of organic compound = 0.32 g
Mass of barium sulphate formed = 0.932 g
1 mol of BaSO4 = 1 mol of S
i.e., 233 g of BaSO4 = 32 g of S
0.932g of BaSO4 = \(\frac{32}{233}\) × 0.932
0.32g of the compound contain = \(\frac{32}{233}\) × 0.932
100g of the compound will contain = \(\frac{32}{233}\) × \(\frac{0.932}{0.32}\) × 100 = 40 %
![]()
Question 54.
0.12 g of an organic compound containing phosphorous gave 0.22 gram Mg2P2O7 by the usual analysis. Calculate the percentage of phosphorus in the compound.
Answer:
Mass of organic compound = 0.12 g
Mass of Mg2P2O7 = 0.22 g
Now 1 mol of Mg2P2O7 = 2 mol of ‘P’ atom
i.e., 222 g of Mg2P2O7= \(\frac{62}{222}\) g of P
0.22 g of Mg2P2O7 = \(\frac{62}{222}\) × 0.22 g of P
0.12 g of the compound contain = \(\frac{62}{222}\) × 0.22 g of P
100 g of the compound contain = \(\frac{62}{222}\) × \(\frac{0.22}{0.12}\) × 100 = 51.20 %
Question 55.
What is the need for purification of an organic compound?
Answer:
In order to study the structure, physical properties, chemical properties and biological properties of organic compounds they must be in the pure state. Hence, to get the compound in pure state, organic compound have to be purified.
![]()
Question 56.
Mention the principle invoked in the separation and purification of organic compound by the following methods. (i) Crystallisation,
(ii) Sublimation,
(iii) Distillation,
(iv) Fractional distillation,
(v) Steam
distillation,
(vi) Azeotropic distil laLion and
(vii) Chromatography.
Answer:
(i) Crystallisation:
It is used to purif’ organic solids by dissolving them in a suitable solvent, followed by filtration of the hot solution and cooling down to separate the solid as crystals.
(ii) Sublimation:
It is a process which a solid is directly converted to its vapour statewithout passing through the liquid state. It is useful to separate volatile and a non volatile solid. eg: dimphor, Benzoic acid, Naphthalene etc.
(iii) Distillation:
This method is to purify liquids from non-volatile impurities, and used for separating the constituents of a liquid mixture which differ in their boiling points.
![]()
(iv) Fractional distillation:
The process of separation of the components in a liquidmixture at their respective boiling points in the form of vapours and the subsequent condensation of those vapours is called fractional distillation.
(v) Steam distillation:
It is applied for the separation and purification of liquids which is appreciably volatile in steam, partially immiscible with water but the impurities are non volatile.
(vi) Azeotropic distillation:
Azeotropes are constant boiling mixtures which distil as a single compound at a fixed temperature.
eg: Ethanol and water mixture in the ratio 95.87 : 4.13. In this type of distillation a third component like C6H6, CCl4, ether, glycerol, glycol is added which act as a depressing agent and depress the partial pressure one component so that the boiling point of that component is raised sufficiently and thus the other component will distil over.
(vii) Chromatography:
Chromatography is defined as a technique for the separation of a mixture brought about by differential movement of the individual compound through porous medium under the influence of moving solvent.
Question 57.
Suggest a method to purify
(i) Camphor containing traces of common salt,
(ii) Kerosene oil containing water,
(iii) A liquid which decomposed its boiling point.
Answer:
(i) Sublimation:
Camphor sublimes white common salt remains as a residue in the China dust.
(ii) The two liquids are immiscible. They can be separated by using a separating funnel. Kerosene being lighter than water forms the upper layer, while water forms the lower layer. The lower layer of water is run off white kerosene oil is obtained. It is dried over anhydrous CaCl2 or MgCl2 and distilled to give pure kerosene.
(iii) Distillation under reduced pressure:
Since, the boiling point of the liquid depends on the pressure acting on it, the boiling point of the liquid is lowered when the pressure is lowered. Thus, a liquid which decomposes at its boiling point can be purified safely at a lower temperature.
![]()
Question 58.
A mixture contains two components A and B. The solution of A and B in water near their boiling points are 10 gram per 100 mL and 2 gram per 100 mL respectively. How will youseparate A and B from the mixtures?
Answer:
Fractional crystallization, when the saturated hot solution is allowed to cool the less soluble substance, B crystallises first leaving the more soluble component A in the mother liquor. Crystals of B are separated by filtration and on further cooling crystals of A appear and can be seperated.
Question 59.
Suggest methods of separation of the following mixtures.
(i) A mixture of liquid (A) (boiling point 365 K) and liquid B (boiling point 345 K).
(ii) A mixture of liquid (C) (boiling point 353 K) and liquid D (boiling point 413 K).
Answer:
(i) Fractional Distillation
(ii) Simple Distillation
Question 60.
The Rf values of A and B in a mixture by TLC in a solvent mixture are 0.65 and 0.42 respectively. 1f the mixture is separated by: column chromatography using the same solvent mixture as a mobile phase which of the two components A or B will elude first. Explain.
Answer::
Since, Rf value of A is 0.65, it is less strongly adsorbed as compared to component B with Rf. value of 0.42. Therefore, on extraction of the column A will elude first.
![]()
Question 61.
How will you separate a mixture of urea and sodium chloride?
Answer:
Both urea and sodium chloride are soluble in water but urea is soluble in alcohol but sodium chloride is insoluble in alcohol. The mixture is shaken with alcohol, when urea goes into the solution while sodium chloride remains undissolved. The sodium chloride is separated by filtration, evaporation of the filtrate gives urea. on further cooling crystals of A appear and can be separated.
Choose the correct answer:
Question 1.
A compound with molecular formula C4H40 has all the four carbon atoms and the oxygen atom in the ring. It also has double bonds. The compound is:
(a) Homocyclic and aromatic
(b) Heterocyclic and aromatic
(e) Homocyclic but not aromatic
(d) Heterocyclic but not aromatic
Answer:
(b) Heterocyclic and aromatic
Hint:
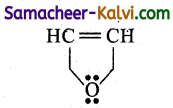
The five membered ring contains an oxygen atom in the ring. Hence, Heterocyclic. It is also aromatic because the pi electrons are delocalised.
Question 2.
Which of the following statement is incorrect?
The members of homologous series of alkanes:.
(a) are all straight chain compounds
(b) have the general molecular formula CnH2n + 2
(c) have similar chemical properties
(d) show regular gradation of physical properties.
Answer:
(a) are all straight chain compounds
Hint:
Alkanes have straight chain and branched chain structures.
![]()
Question 3.
The enolic form of ethyl aceto acetate is shown below has:
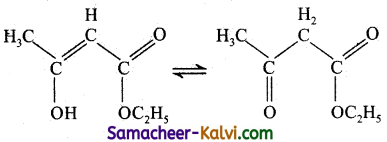
(a) 9 sigma bonds and two pi bonds
(b) 9 sigma bonds and one pi bond
(c) 18 sigma bonds and two pi bonds
(d) 16 sigma bonds and one pi bond
Answer:
(c) 18 sigma bonds and two pi bonds
Hint:
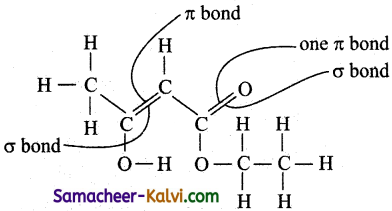
no. of σ bonds = 18
no. of π bonds = 2
Question 4.
Considering the state of hybridisation of carbon atom, find out the molecule among the following which is linear?
(a) CH3CH2CH2CH3
(b) CH3 CH=CH.CH3
(c) CH3C ≡ C.CH3
(d) CH2 = CH.CH2C ≡ CH
Answer:
(c) CH3C ≡ C.CH3
Hint:
All carbon atoms are sp3 hybridised – tetrahedral.
(b) It is an alkene. Contain a double bond sp2 hybridised – plane triangle,
(c) It is an alkyne. sp hybridised – linear.
(d) It contains a double and triple bonded carbon sp2 and sp hybridised.
![]()
Question 5.
The correct IUPAC name of CH3CH2CH(CH3) CH(C2H5)2 is: ;
(a) 4 – Ethyl – 3- methyl hexane
(b) 3- Ethyl – 4 – methyl hexane
(c) 4 Methyl – 3 – ethyl hexane
(d) 2, 3 dimethyl pentane
Answer:
(b) 3- Ethyl – 4 – methyl hexane
Hint:
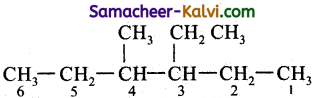
Question 6.
Which of the following structures represents 2,2,3, trimethyl hexane?
(a) CH3C (CH3)2 CH2 CH2 CH . (CH3)2
(b) CH3CH (CH3) CH2 CH (CH3) CH2 CH3
(c) CH3 C (CH3)2 CH (CH3) CH2 CH2 CH3
(d) CH3 C (CH3)2 CH2 C(CH3)2 CH3
Answer:
(b) CH3CH (CH3) CH2 CH (CH3) CH2 CH3
Hint:
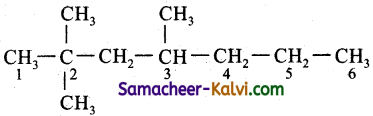
![]()
Question 7.
The IUPAC name of the compound with the structural formula CH3 CH = CH — C = CH is:
(a) Pent – 2 – en – 4 yne
(b) Pent – 1 – yn – 3 ene
(c) Pent – 3 – en – 1 yne
(d) Pent – 2 – en – 5yne
Answer:
(c) Pent – 3 – en – 1 yne
Hint:
While naming unsaturated hydrocarbons the locant of the double bond or the triple bond
is place before the suffix ‘ene’ or ‘yne’.
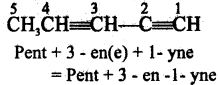
Question 8.
The IUPAC name of the following compound is:
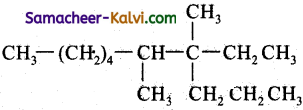
(a) 3, 4, Dimethyl – 3 – n – propyl octane
(b) 6, 7, Dimethyl – 2 – n – propyl octane
(c) 6, 7, Dimethyl – 7 – ethyl decane
(d) 4 – Ethyl- 4, 5 dimethyl decane
Answer:
(d) 4 – Ethyl- 4, 5 dimethyl decane
Hint:
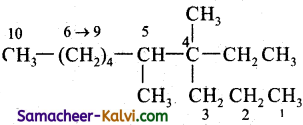
Question 9.
An organic compound contains
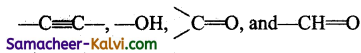
groups. Which of these will be considered as the principal functional group while giving
(a) — C ≡ C —
(b) — OH
(c) ![]()
(d) — CH = O
Answer:
(d) — CH = O
Hint:
The order of preference is aldehyde, ketones, alcohols, alkynes.
![]()
Question 10.
Given
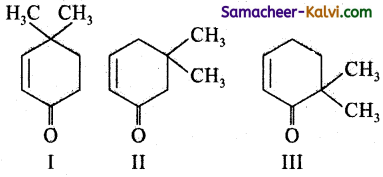
Which of the following compounds can exhibit tautomerism?
(a) II and III
(b) I, II and III
(c) I and II
(d) I and III
Answer:
(b) I, II and III
Hint:
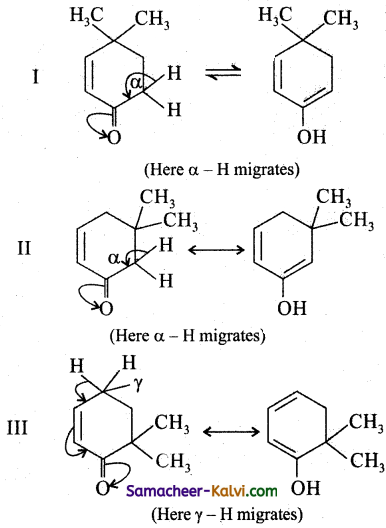
Keto enol tautomerism involves migration ofa hydrogen atom either from a (1, 3) or γ (1, 5)or ∈ (1, 7) position to the oxygen atom of the keto group.
![]()
Thus I, II abd III all will exhibit tautomerism.
Question 11.
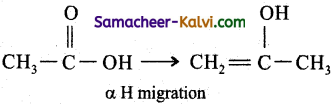
Structures CH3COOH and CH2 = CH.(OH).CH3 represent:
(a) metamerism
(b) functional isomerism
(c) keto-enol tautomerism
(d) position isomerism
Answer:
(c) keto-enol tautomerism
Hint:
Question 12.
Fisher projection indicates:
(a) Horizontal substituents above the plane.
(b) Vertical substituents above the plane.
(c) Both horizontal and vertical substituents below the plane.
(d) Both horizontal and vertical substituents above the plane.
Answer:
(a) Horizontal substituents above the plane.
Question 13.
An organic compound which produces a bluish green coloured flame on heating in the presence of copper is:
(a) chlorobenzene
(b) benzaldehyde
(c) aniline
(d) benzoic acid
Answer:
(a) chlorobenzene
Hint:
Any organic compound which contains chlorine when heated in flame in the presence of copper produces a bluish blue flame due to the formation of volatile cupric chloride.
![]()
Question 14.
0.28 g of a nitrogeneous compound was Kjeldahlised to produce 0.17 g of NH3. The percentage of nitrogen in the organic compound is:
(a) 5
(b) 30
(c) 50
(d) 80
Answer:
(c) 50
% N = \(\frac{14}{7}\) × \(\frac{0.17}{0.28}\) × 100 = 50 %
Question 15.
Which of the following structures permit cis-trans isomerism?
(a) X2C = CY2
(b) XYZ = CZ
(c) X2C = CXY
(d) XYC = CXZ
Answer:
(a) X2C = CY2
Hint:
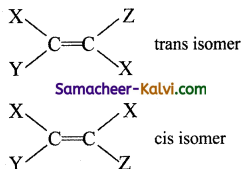
Question 16.
Assertion :
Alkanes containing more than three carbon atoms exhibit chain isomerism.
Reason :
All carbon atoms in alkanes are sp3 hybridised.
(a) Both the assertion and the reason are true and the reason is the correct explanation of assertion.
(b) Both the assertion and reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(c) Assertion is true but reason is false.
![]()
Question 17.
Assertion:
All the carbon atoms in but-2-ene lie in one plane.
Reason :
All the carbon atoms in but-2-ene re sp2 hybridised,
(a) Both the assertion and reason are true andthe reason is the correct explanation of assertion.
(b) Both the assertion and the reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false,
(d) Both assertion and reason are false.
Answer:
(c) Assertion is true but reason is false.
Hint:
![]()
The correct statement for the reason, carbon atoms 2, 3 alone are sp2 hybridised.
Question 18.
Assertion:
Simple distillation can help in separating a mixture of propan- l-ol (boiling point 97°C) and propanone (56°C).
Reason :
Liquids with a difference of more than 20°C in their boiling point can be separated by simple distillation.
(a) Both the assertion and reason are true and the reason is the correct explanation of assertion.
(b Both the assertion and the reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(a) Both the assertion and reason are true and the reason is the correct explanation of assertion.
Question 19.
Assertion:
A sulphur present in an organic compound can be estimated quantitatively by carius method.
Reason :
Sulphur can be easily separated from other atoms in the molecule and gets precipitated as yellow solid.
(a) Both the assertion and reason are true and the reason is the correct explanation of assertion.
(b) Both the assertion and the reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false
(d) Both assertion and reason are false.
Answer:
(a) Both the assertion and reason are true and the reason is the correct explanation of assertion.
Hint:
Sulphur is oxidised to H2SO4 and then estimated as barium sulphate.
![]()
Question 20.
Assertion:
Components of a mixture and red and blue inks can be separated by distributing the components between stationary and mobile phase.
Reason :
The coloured components of inks migrate at different rates because paper selectively retains different components according to difference in their position between two phases,
(a) Both the assertion and the reason are true and the reason is the correct explanation of assertion.
(b) Both the assertion and the reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(a) Both the assertion and the reason are true and the reason is the correct explanation of assertion.
Question 21.
Select the odd man out of the following j represents a homologous series.
(a) ethane, ethylene,ethyne
(b) methane, methanol, methanal
(c) methane, ethane, propane
(d) 1-hexane,2-hexane,3-hexane
Answer:
(c) methane, ethane, propane
Question 22.
In which of the following functional isomerism | is not possible? .
(a) Alcohols
(b) Aldehydes
(c) Alkyl halides
(d) Alkyl cyanides
Answer:
(c) Alkyl halides
![]()
Question 23.
Among the following pairs the pair that ! illustrates functional isomerism is:
(a) 1 – butanol and 2 – butanol
(b) dimethyl ether and ethanol
(c) cis – 2 – butene and trans – 2 – butene
(d) ethanal and ethanol
Answer:
(b) dimethyl ether and ethanol
Question 24.
Consider the following four compounds:
I. CH3CH2CH2CH2CHO
II. CH3CH2CH2COCH3
III. CH3CH2COCH2CH3
IV. 
(i) Which of the following pairs are position isomers?
(a) I and II
(b) II and III
(c) II and IV
(d) III and IV
Answer:
(b) II and III
(ii) Which of the following pairs are not functional ; isomers?
(a) II and III
(b) II and IV
(c) I and II
(d) I and IV
Answer:
(a) and (d)
![]()
Question 25.
Assertion:
Butane and 2 methyl butane are homologous.
Reason :
Butane is a straight chain alkane, while 2 – methyl butane is a branched chain alkane.
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If both assertion and reason are false.
Answer:
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
Question 26.
Assertion:
A mixture of ortho nitro phenol and para nitro phenol can be separatedn by steam distillation.
Reason:
Pata nitro phenol is steam volatile while ortho nitro phenol is not.
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If both assertion and reason are false.
Answer:
(c) If assertion is true but reason is false.
![]()
Question 27.
Assertion:
NH2 — C — NH2 gives red colour in Lassaigne’s test.
Reason :
Compounds having nitrogen and carbon gives red colour in Lassaigne’s test.
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If both assertion and reason are false.
Answer:
(c) If assertion is true but reason is false.
Hint:
Correct Reason: Compounds containing N, S and C give red colour in Lassaigne’s test.
Question 28.
Assertion:
Lassaigne’s test is not shown by diazonium salts.
Reason :
Diazonium salts lose N2 on heating much before they have a chance to react with fused sodium.
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If both assertion and reason are false.
Answer:
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
![]()
Question 29.
Assertion:
All the carbon atoms of but-2-ene lie in one plane.
Reason :
All the carbon atoms in but-2-ene are sp2 hybridised.
(a) If both assertion and reason are correct and reason is the correct explanation of the assertion.
(b) If both assertion and reason are true but reason is not the correct explanation of the assertion.
(c) If assertion is true but reason is false.
(d) If both assertion and reason are false.
Answer:
(c) If assertion is true but reason is false.
Question 30.
Choose the correct statement from the following:
(a) Cyclo butane is an aromatic compound.
(b) Naphthalene is a hetero cyclic compound.
(c) Pyrole is a non-benzenoid compound.
(d) Thiophene is a homocyclic compound.
Answer:
(c) Pyrole is a non-benzenoid compound.
![]()
Question 31.
Which of the following statement is correct with respect to alicyclic compound?
I. They are closed chain compounds.
II. Cyclo propane is an alicyclic compound.
III. Benzene is also an alicyclic compound.
(a) I and II
(b) II and III
(c) III only
(d) I and III
Answer:
(a) I and II
Question 32.
Choose the incorrect statement from the following:
(a) Pent – 1 – ene and pent – 2 – ene are position isomers.
(b) Propanal and propanone are functional isomers.
(c) Methyl propyl ether and diethyl ether are metamers.
(d) Pentan- 2 – one and pentan – 3 – one are position isomers.
Answer:
(d) Pentan-2 – one and pentan – 3 – one are position isomers.
Hint:
They are metamers.
![]()
Question 33.
Choose the incorrect statement from the following:
(a) Camphor containing traces of common salt can be purified by sublimation.
(b) Kerosene oil containing water can be separated by using a separating funnel.
(c) A mixture of liquid A(b.pt 365 K) and liquid B(356 K) can be separated by fractional distillation.
(d) A mixture of water (b.pt 373 K) and aniline (b.pt 457 K) at 760 mm can be separated by fractional distillation.
Answer:
(d) A mixture of water (b.pt 373 K) and aniline (b.pt 457 K) at 760 mm can be separated by fractional distillation.
Hint:
They are separated by steam distillation.
Question 34.
Which of the following statement is incorrect with respect to cis-trans isomerism?
(a) It arises due to restricted rotation aeron C=C double bond.
(b) They have different physical properties.
(c) The ‘cis’ isomer is more polar than the ‘trans’ isomer.
(d) The ‘trans’ isomer is less stable than the ‘cis’ isomer.
Answer:
(d) The ‘trans’ isomer is less stable than the ‘cis’ isomer.
![]()
Question 35.
Match the entities of column I with appropriate entities of column II.
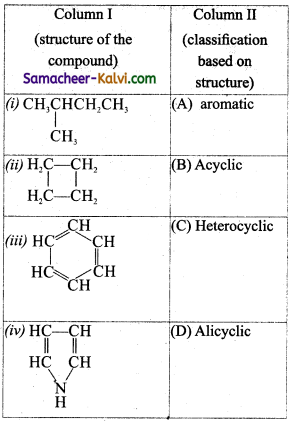
(a) (i) – (B), (ii) – (D), (iii) – (A), (iv) – (C)
(b) (i) – (D), (ii) – (B), (iii) – (C), (iv) – (A)
(c) (i) – (B), (ii) – (A), (iii) – (D), (iv) – (C)
(d) (i) – (D), (ii) – (C), (iii) – (A), (iv) – (B)
Answer:
(a)(i) – (B), (ii) – (D), (iii) – (A), (iv) – (C)
![]()
Question 36.
Match the entities of column I with appropriate entities of column II.
| Column I | Column II |
| (i) Two solids have different solubilities in a solvent and which do not undergo reaction when dissolved | (A) Simple distillation |
| (ii) Liquid that decomposes at its boiling point | (B) Crystallisation |
| (Hi) Two volatile liquids which have boiling points close to each other. | (C) Distillation under reduced pressure |
| (iv) Two liquids which have large difference in their boiling points. | (D) Fractional distillation |
(a) (i) – (B), (ii) – (D), (iii) – (A), (iv) – (C)
(b) (i) – (B), (ii) – (C), (iii) – (D), (iv) – (A)
(c) (i) – (C), (ii) – (D), (iii) – (B), (iv) – (A)
(d) (i) – (B), (ii) – (D), (iii) – (C), (iv) – (A)
Answer:
(b) (i) – (B), (ii) – (C), (iii) – (D), (iv) – (A)