Tamilnadu State Board New Syllabus Samacheer Kalvi 11th Chemistry Guide Pdf Chapter 3 Periodic Classification of Elements Text Book Back Questions and Answers, Notes.
Tamilnadu Samacheer Kalvi 11th Chemistry Solutions Chapter 3 Periodic Classification of Elements
11th Chemistry Guide Periodic Classification of Elements Text Book Back Questions and Answers
Textual Questions:
I. Choose the best Answer:
Question 1.
What would be the IUPAC name for an element with atomic number 222?
(a) bibibiium
(b) bididium
(c) didibium
(d) bibibium
Answer:
(d) bibibium
Question 2.
The electronic configuration of the elements A and B are 1s2, 2s2, 2p6, 3s2 and 1s2, 2s2, 2p5 respectively. The formula of the ionic compound that can be formed between these elements is
(a) AB
(b) AB2
(c) A2B
(d) none of the above
Answer:
(b) AB2
Question 3.
The group of elements in which the differentiating electron enters the anti penultimate shell of atoms are called
(a) p-block elements
(b) d-block elements
(c) s-block elements
(d) f-block elements
Answer:
(d) f-block elements
Question 4.
In which of the following options the order of arrangement does not agree with the variation of property indicated against it?
(a) I < Br < Cl < F (increasing electron gain enthalpy)
(b) Li < Na < K < Rb (increasing metallic radius)
(c) Al3+ < Mg2+ < Na+ < F– (increasing ionic size)
(d) B < C < O < N (increasing first ionisation enthalpy)
Answer:
(a) I < Br < Cl < F (increasing electron gain enthalpy)
Question 5.
Which of the following elements will have the highest electronegativity?
(a) Chlorine
(b) Nitrogen
(c) Cesium
(d) Fluorine
Answer:
(d) Fluorine
![]()
Question 6.
Various successive ionisation enthalpies (in kjmol 1) of an element are given below.
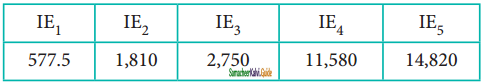
The element is
(a) phosphorus
(b) Sodium
(c) Aluminium
(d) Silicon
Answer:
(c) Aluminium
Question 7.
In the third period the first ionization potential is of the order.
(a) Na > Al > Mg > Si > P
(b) Na < Al < Mg < Si < P
(c) Mg > Na > Si > P > Al
(d) Na < Al < Mg < P < Si
Answer:
(b) Na < Al < Mg < Si < P
Question 8.
Identify the wrong statement.
(a) Amongst the isoelectronic species, smaller the positive charge on cation, smaller is the ionic radius
(b) Amongst isoelectric species greater the negative charge on the anion, larger is the ionic radius
(c) Atomic radius of the elements increases as one moves down the first group of the periodic table
(d) Atomic radius of the elements decreases as one moves across from left to right in the 2nd period of the periodic table.
Answer:
(a) Amongst the isoelectronic species, smaller the positive charge on cation, smaller is the ionic radius
Question 9.
Which one of the following arrangements represent the correct order of least negative to most negative electron gain enthalpy
(a) Al < O < C < Ca < F
(b) Al < Ca < O < C < F
(c) C < F < O < Al < Ca
(d) Ca < Al < C < O < F
Answer:
(d) Ca < Al < C < O < F
Question 10.
The correct order of electron gain enthalpy with negative sign of F, Cl, Br and I having atomic number 9, 17, 35 and 53 respectively is
(a) I > Br > Cl > F
(b) F > Cl > Br > I
(c) Cl > F > Br > I
(d) Br > I > Cl > F
Answer:
(c) Cl > F > Br > I
![]()
Question 11.
Which one of the following is the least electronegative element?
(a) Bromine
(b) Chlorine
(c) Iodine
(d) Hydrogen
Answer:
(d) Hydrogen
Question 12.
The element with positive electron gain enthalpy is
(a) Hydrogen
(b) Sodium
(c) Argon
(d) Fluorine
Answer:
(c) Argon
Question 13.
The correct order of decreasing electronegativity values among the elements X, Y, Z and A with atomic numbers 4, 8, 7 and 12 respectively
(a) Y > Z > X > A
(b) Z > A > Y > X
(c) X > Y > Z > A
(d) X > Y > A > Z
Answer:
(a) Y > Z > X > A
Question 14.
Assertion:
Helium has the highest value of ionisation energy among all the elements known
Reason:
Helium has the highest value of electron affinity among all the elements known
(a) Both assertion and reason are true and reason is correct explanation for the assertion
(b) Both assertion and reason are true but the reason is not the correct explanation for the assertion
(c) Assertion is true and the reason is false
(d) Both assertion and the reason are false
Answer:
(c) Assertion is true and the reason is false
Question 15.
The electronic configuration of the atom having | maximum difference in first and second ionisation j energies is
(a) 1s2, 2s2, 2p6, 3s1
(b) 1s2, 2s2, 2p6, 3S2
(c) 1s2, 2s2, 2p6, 3s2, 3s2, 3p6, 4s1
(d) 1s2, 2s2, 2p6, 3s2, 3p1
Answer:
(a) 1s2, 2s2, 2p6, 3s1
![]()
Question 16.
Which of the following is second most electronegative element?
(a) Chlorine
(b) Fluorine
(c) Oxygen
(d) Sulphur
Answer:
(a) Chlorine
Question 17.
IE1 and IE2 of Mg are 179 and 348 kcal mol-1 respectively. The energy required for the reaction Mg → Mg2+ + 2e– is
(a) + 169 kcal mol-1
(b) -169 kcal mol-1
(c) +527 kcalmol-1
(d) -527 kcal mol-1
Answer:
(c) +527 kcalmol-1
Question 18.
In a given shell the order of screening effect is
(a) s > p > d > f
(b) s > p > f > d
(c) f > d > p > s
(d) f > p > s > d
Answer:
(a) s > p > d > f
Question 19.
Which of the following orders of ionic radii is correct?
(a) H > H+ > H
(b) Na+ > F– > O2-
(c) F > O2- > Na+
(d) None of these
Answer:
(d) None of these
Question 20.
The First ionisation potential of Na, Mg and Si are 496, 737 and 786 kJ mol-1 respectively. The ionisation potential of Al will be closer to
(a) 760 kJ mol-1
(b) 575 kJ mol-1
(c) 801 kJ mol-1
(d) 419 kJ mol-1
Answer:
(b) 575 kJ mol-1
![]()
Question 21.
Which one of the following is true about metallic character when we move from left to right in a period and top to bottom in a group?
(a) Decreases in a period and increases along the group
(b) Increases in a period and decreases in a group
(c) Increases both in the period and the group
(d) Decreases both in the period and in the group
Answer:
(b) Increases in a period and decreases in a group
Question 22.
How does electron affinity change when we move from left to right in a period in the periodic table?
(a) Generally increases
(b) Generally decreases
(c) Remains unchanged
(d) First increases and then decreases
Answer:
(a) Generally increases
Question 23.
Which of the following pairs of elements exhibit diagonal relationship?
(a) Be and Mg
(b) Li and Mg
(c) Be and B
(d) Be and Al
Answer:
(d) Be and Al
![]()
II. Write brief answer to the following questions:
Question 24.
Define modern periodic law.
Answer:
The modem periodic law states that “The physical and chemical properties of the elements are a periodic function of their atomic numbers.”
Question 25.
What are isoelectronic ions? Give examples.
Answer:
Two ions having the same number of electrons are called isoelectronic ions.
Example: Na+ (1s2 2s2 2p6) and F– (1s2 2s2 2p6). Both these ions contain eight electrons.
Question 26.
What is an effective nuclear charge?
Answer:
The net nuclear charge experienced by valence electrons in the outermost shell is called the effective nuclear charge.
Zeff = Z – S
Where Z = Atomic number
S = Screening constant calculated by using Slater’s rules.
Question 27.
Is the definition given below for ionization enthalpy is correct?
Answer:
“Ionisation enthalpy is defined as the energy required to remove the most loosely bound electron from the valence shell of an atom”
The given statement is not correct. Ionization energy is defined as the minimum amount of energy required to remove the most loosely bound electron from the valence shell of the isolated neutral gaseous atom in its ground state.
Question 28.
Magnesium loses electrons successively to form Mg+, Mg2+, and Mg3 ions. Which step will have the highest ionization energy and why?
Answer:
Magnesium loses electrons successively in the following steps,
Step 1:
Mg(g) + IE1 → Mg+(g) + 1e, Ionisation energy = I.E1
Step 2:
Mg+(g)+ IE2 → Mg2+(g) + 1e, Ionisation energy = I.E2
Step 3:
Mg2+(g) + IE3 → Mg3+(g) + 1e, Ionisation energy = I.E3
The total number of electrons is less in the cation than the neutral atom while the nuclear charge remains the same. Therefore, the effective nuclear charge of the cation is higher than the corresponding neutral atom. Thus, the successive ionization energies, always increase in the following order I.E1 < I.E2 < I.E3. Thus, Step-3 will have the ionization energy.
Question 29.
Define electronegativity.
Answer:
Electronegativity is defined as the relative tendency of an element present in a covalently bonded molecule, to attract the shared pair of electrons towards itself.
Question 30.
How would you explain the fact that the second ionisation potential is always higher than the first ionisation potential?
Answer:
- The second ionization potential is always higher than the first ionization potential.
- Removal of one electron from the valence orbit of a neutral gaseous atom is easy so first ionization energy is less. But from a uni positive ion, removal of one more electron becomes difficult due to the more forces of attraction between the excess of protons and less number of electrons.
- Due to greater nuclear attraction, second ionization energy is higher than first ionization energy.
![]()
Question 31.
The energy of an electron in the ground state of the hydrogen atom is -2.8 × 10-8 J. Calculate the ionization enthalpy of atomic hydrogen in terms of kJ mol-1
Answer:
Energy of an electron in the ground state of the hydrogen atom is – 2.8 × 10-8.
The ionization energy of atomic hydrogen is 2.8 × 10-18 × 6.023 × 1023 J/mol
= 16.86 × 105 J/mol
= 1686 kJ/mol.
Question 32.
The electronic configuration of an atom is one of the important factors which affects the value of ionization potential and electron gain enthalpy. Explain.
Answer:
- The electronic configuration of an atom affects the value of ionization potential and electron gain enthalpy.
- Half-filled valence shell electronic configuration and completely filled valence shell electronic configuration are more stable than partially filled electronic configuration.
- For e.g. Beryllium (Z = 4) 1s2 2s2 (completely filled electronic configuration)
Nitrogen (Z = 7) 1s2 2s2 2px1 2py1 2pz1 (half-filled electronic configuration) Both beryllium and nitrogen have high ionization energy due to more stable nature. - In the case of beryllium (1s2 2s2), nitrogen (1s2 2s2 2p3) the addition of extra electrons will disturb their stable electronic configuration and they have almost zero electron affinity.
- Noble gases have stable ns2 np6 configuration and the addition of further electrons is unfavorable and they have zero electron affinity.
Question 33.
In what period and group will an element with Z = 118 will be present?
Answer:
The element Ununoctium (Oganesson, Z – 118) present in the 7th period and 18th group of the periodic table.
Question 34.
Justify that the fifth period of the periodic table should have 18 elements on the basis of quantum numbers.
Answer:
The fifth period of the periodic table has 18 elements. 5th period starts from Rb to Xe (18 elements).
5th period starts with principal quantum number n = 5 and l = 0, 1, 2, 3 and 4.
When n = 5, the number of orbitals = 9.
1 for 5s
5 for 4d
3 for 5p
The total number of orbitals = 9.
Total number of electrons that can be accommodated in 9 orbitals = 9 x 2 = 18.
Hence the number of elements in the 5th period is 18.
![]()
Question 35.
Elements a, b, c and d have the following electronic configurations:
a: 1s2, 2s2, 2p6
b: 1s2, 2s2, 2p6, 3s2, 3p1
c: 1s2, 2s2, 2p6, 3s2, 3p6
d: 1s2, 2s2, 2p1
Which elements among these will belong to the same group of the periodic table?
Answer:
The elements ‘a’ [Ne (Z= 10), 1s2, 2s2, 2p6] and ‘c’ [Ar (Z = 18), 1s2, 2s2, 2p6, 3s2, 3p6] have the same valence electronic configuration and hence, belongs to the same group, i.e., group 18 of the periodic table . Similarly, the elements ‘b’ [ Al (Z = 13), 1s2, 2s2, 2p6, 3s2, 3p1] and ‘d’ [B (Z= 5), 1s2, 2s2, 2p1] have the same valence electronic configuration and hence, belongs to the same group, i.e., group 13 of the periodic table.
Question 36.
Give the general electronic configuration of lanthanides and actinides?
Answer:
- The electronic configuration of lanthanides is 4f1-14 5d0-16s2.
- The electronic configuration of actinides is 5f1-14 6d0-1 7s2.
Question 37.
Why do halogens act as oxidizing agents?
Answer:
Halogens are having the general electronic configuration of ns2, np5 and readily accept an electron to get the stable noble gas electronic configuration. Therefore, halogens have high electron affinity. Hence, halogens act as oxidizing agents.
Question 38.
Mention any two anomalous properties of second-period elements.
Answer:
- In the 1st group, lithium forms compounds with more covalent character while the other elements of this group form only ionic compounds.
- In the 2nd group, beryllium forms compounds with more covalent character while the other elements of this family form only ionic compounds.
Question 39.
Explain the Pauling method for the determination of ionic radius.
Answer:
Ionic radius is defined as the distance from the centre of the nucleus of the ion upto which it exerts its influence on the electron cloud of the ion. The ionic radius of a uni-univalent crystal can be calculated using Pauling’s method from the interionic distance between the nuclei of the cation and anion. Pauling assumed that ions present in a crystal lattice are perfect spheres, and they are in contact with each other and therefore,
d = rc+ + rA- …………..(1)
where ‘d’ is the distance between the centre of the nucleus of the cation C+ and A–. r c+ and rA- are the radius of the cation and anion respectively.
Pauling also assumed that the radius of the ion having noble gas electronic configuration (Na+ and Cl– having 1s2, 2s2, 2p6 configuration) is inversely
proportional to the effective nuclear charge felt at the periphery of the ion.
i.e., rc+ ∝ \(\frac{1}{\left(Z_{e f f}\right)^{C+}}\) ………(2)
and rA- ∝ \(\frac{1}{\left(Z_{e f f}\right)^{A-}}\) ………….(3)
where Zeff is the effective nuclear charge.
Zeff = Z – S.
Dividing the equation (2) by (3)
\(\frac{r_{c^{+}}}{r_{A^{-}}}=\frac{\left(Z_{e f f}\right)^{A-}}{\left(Z_{e f f}\right)^{C+}}\)
On solving the equations (1) and (4), the ionic radius of cation and anion are calculated.
![]()
Question 40.
Explain the periodic trend of ionisation potential.
Answer:
(a) The energy required to remove the most loosely held electron from an isolated gaseous atom is called ionization energy.
(b) Variation in a period:
Ionization energy is a periodic property. On moving across a period from left to right, the ionization enthalpy value increases. This is due to the following reasons.
- Increase of nuclear charge in a period
- Decrease of atomic size in a period
Because of these reasons, the valence electrons are held more tightly by the nucleus. Therefore, ionization enthalpy increases.
(c) Variation in a group:
As we move from top to bottom along a group, the ionization enthalpy decreases. This is due to the following reasons.
- A gradual increase in atomic size
- Increase of screening effect on the outermost electrons due to the increase of the number of inner electrons.
Hence, ionization enthalpy is a periodic property.
Question 41.
Explain the diagonal relationship.
Answer:
On moving diagonally across the periodic table, the second and third-period elements show certain similarities. Even though the similarity is not the same as we see in a group, it is quite pronounced in the following pair of elements.
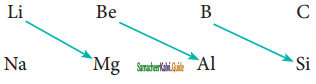
The similarity in properties existing between the diagonally placed elements is called ‘diagonal relationship’.
Question 42.
Why the first Ionisation enthalpy of sodium is lower than that of magnesium while its second ionisation enthalpy Is higher than that of magnesium?
Answer:
The 1st ionization enthalpy of magnesium is higher than that of Na due to the higher nuclear charge and slightly smaller atomic radius of Mg than Na. After the loss of the first electron, Na+ formed has the electronic configuration of neon (2, 8). The higher stability of the completely filled noble gas configuration leads to a very high second ionization enthalpy for sodium. On the other hand, Mg+ formed after losing the first electron still has one more electron in its outermost (3s) orbital. As a result, the second ionization enthalpy of magnesium is much smaller than that of sodium.
![]()
Question 43.
By using Pauling’s method calculate the ionic radii of K+ and Cl– ions in the potassium chloride crystal. Given that dK + – Cl– = 3.14 Å.
Answer:
d = rK+ + rCl- = 3.14 Å
\(\frac{r_{K+}}{r_{C l-}}=\frac{\left(Z_{e f f}\right)^{C l-}}{\left(Z_{e f f}\right)^{K+}}\)
(Zeff)Cl- = Z – S = 17 – 10.9 = 6.1
(Zeff)K+ = Z – S = 19 – 16.8 = 2.2
\(\frac{r_{K+}}{r_{C l-}}=\frac{6.1}{2.2}\) = 2.77
rK+ = 2.77 rCl-
2.77 rCl- + rCl- = 3.17 Å
3.77 rCl- = 3.17 Å
rCl- = 0.83 Å
rK+ = (3.14 – 0.83) Å = 2.31 Å
The ionic radius of the K+ ion is 2.31 Å and Cl– ion is 0.83 Å.
Question 44.
Explain the following, give appropriate reasons.
(i) Ionisation potential of N is greater than that of O:
Answer:
Nitrogen with 1s2, 2s2, 2p3 electronic configuration has higher ionization energy than oxygen. Since the half-filled electronic configuration is more stable, it requires higher energy to remove an electron from the 2p orbital of nitrogen. Whereas the removal of one 2p electron from oxygen leads to a stable half-filled configuration. This makes it comparatively easier to remove 2p electron from oxygen.
(ii) First ionisation potential of the C-atom is greater than that of the B atom, whereas the reverse is true is for the second ionisation potential.
Answer:
The first ionization potential of the C atom and B-atom are as follows:
C(1s2, 2s2, 2p2) + IE1 → C+ (1s2, 2s2, 2p1)
B(1s2, 2s2, 2p1) + IE1 → B+ (1s2, 2s2)
The ionization energy usually increases along a period. Hence, the first ionization energy of carbon is greater than that of Boron.
The second ionization potential of the C atom and B atom is as follows:
C+ (1s2, 2s2, 2p1) + IE2 → C+(1s2, 2s2)
B+ (1s2, 2s2) + IE2 → B+ (1s2, 2s1)
B+ has completely filled 2s orbital which is more stable than the partially filled valence shell electronic configuration of the C+ atom. Hence, the second ionization energy of Boron is greater than that of carbon.
(iii) The electron affinity values of Be, Mg, and noble gases are zero, and those of N (0.02 eV) and P (0.80 eV) are very low.
Answer:
Be, Mg and noble gases have completely filled stable configuration and the addition of further electron is unfavourable and requires energy. The addition of extra electrons will disturb their stable electronic configuration and hence, they have almost zero electron affinity.
Nitrogen and Phosphorus have a half-filled stable configurations and the addition of further electrons is unfavourable and requires energy. The addition of extra electrons will disturb their stable electronic configuration and hence, they have very low zero electron affinity.
(iv) The formation of from F–(g) from F(g) is exothermic while that of O2-(g) from O(g) is endothermic.
Answer:
The sizes of oxygen and fluorine atoms are comparatively small and they have high electron density. The extra electron added to fluorine has to accommodate in the 2p orbital which is relatively compact. Hence, the formation of F- from F is exothermic. In the case of oxygen, the formation of O2- from O is endothermic due to extra stability of the completely filled 2p orbital of O2- formation.
![]()
Question 45.
What is the screening effect?
Answer:
The repulsive force between inner shell electrons and the valence electrons leads to a decrease in the electrostatic attractive forces acting on the valence electrons by the nucleus. Thus the inner shell electrons act as a shield between the nucleus and the valence electrons. This effect is called the shielding effect (or) screening effect.
Question 46.
Briefly give the basis for Pauling’s scale of electronegativity.
Answer:
Pauling’s scale:
- Electronegativity is the relative tendency of an element present in a covalently bonded molecule to attract the shared pair of electrons towards itself.
- Pauling assigned arbitrary values of electronegativities for hydrogen and fluorine as 2.2 and 4, respectively.
- Based on this the electronegativity values for other elements can be calculated using the following expression.
(XA-XB) = 0.182 √EAB – (EAA EBB)
Where EAB , EAA, and EBB are the bond dissociation energies of AB, A2, and B2 molecules respectively.
XA and XB are electronegativity values of A and B.
Question 47.
State the trends in the variation of electronegativity in groups and periods.
Answer:
Variation of Electronegativity in a period:
The electronegativity generally increases across a period from left to right. As discussed earlier, the atomic radius decreases in a period, as the attraction between the valence electron and the nucleus increases. Hence, the tendency to attract shared pair of electrons increases. Therefore, electronegativity also increases in a period.
Variation of Electronegativity in a group:
The electronegativity generally decreases down a group. As we move down a group, the atomic radius increases, and the nuclear attractive force on the valence electron decreases. Hence, the electronegativity decreases. Noble gases are assigned zero electronegativity. The electronegativity values of the elements of 5-block show the expected decreasing order in a group. Except for 13th and 14th groups, all other p-block elements follow the expected decreasing trend in electronegativity.
![]()
11th Chemistry Guide Periodic Classification of Elements Additional Questions and Answers
I. Very Short Question and Answers (2 Marks):
Question 1.
State periodic law.
Answer:
Periodic law states that ‘‘the properties of the elements are the periodic functions of their atomic weights”.
Question 2.
Write a note about Chancourtois classification.
Answer:
In this system, elements that differed from each other in atomic weight by 16 or multiples of 16 fell very nearly on the same vertical line. Elements lying directly under each other showed a definite similarity. This was the first periodic law.
Question 3.
What is Lavoiser’s classification of elements?
Answer:
Lavoiser classified the substances into four groups of elements namely acid-making elements, gas-like elements, metallic elements, and earthy elements.
Question 4.
State Mendeleev’s periodic law.
Answer:
This law states that “The physical and chemical properties of elements are a periodic function of their atomic weights.”
Question 5.
What are groups and periods?
Answer:
All the elements are arranged in the modem periodic table which contains 18 vertical columns and 7 horizontal rows. In the periodic table, the vertical columns are called groups, and horizontal rows are called periods.
![]()
Question 6.
State modern periodic law.
Answer:
The modem periodic law states that, “The physical and chemical properties of the elements are periodic function of their atomic numbers.”
Question 7.
What are p-block elements? Give their general electronic configuration.
Answer:
The elements of groups 13 to 18 are called p-block elements or representative elements and have a general electronic configuration ns2, np1 – 6.
Question 8.
Mention the names of the elements with atomic numbers 101, 102, 109, and 110.
Answer:
Z = 101 IUPAC name : Mendelevium
Z = 102 IUPAC name : Nobelium
Z = 109 IUPAC name : Meitnerium
Z = 110 IUPAC name : Darmstadtium
Question 9.
What are f-block elements? Give their properties.
Answer:
The lanthanides and the actinides are called f-block elements. These elements are metallic in nature and have high melting points. Their compounds are mostly coloured. These elements also show variable oxidation states.
Question 10.
Give the name and electronic configuration of elements of group and 2 groups.
Answer:
- Elements of 1st group are called alkali metals. Their electronic configuration is ns1.
- Elements of 2nd group are called alkaline earth metals. Their electronic configuration is ns2.
![]()
Question 11.
Define Atomic radius.
Answer:
The atomic radius of an atom is defined as the distance between the centre of its nucleus and the outermost shell containing the valence electron.
Question 12.
Write any two characteristic properties of alkaline earth metals.
Answer:
- Alkaline earth metals readily lose their outermost electrons to form a +2 ion.
- As we go down the group. their metallic character and reactivity are increased.
Question 13.
Why is the covalent radius shorter than the actual atomic radius?
Answer:
The formation of a covalent bond involves the overlapping of atomic orbitals and it reduces the expected internuclear distance. Therefore, the covalent radius is always shorter than the actual atomic radius.
Question 14.
Define metallic radius.
Answer:
Metallic radius is defined as one-half of the distance between two adjacent metal atoms in the closely packed metallic crystal lattice.
Question 15.
Halogens and chalcogens have highly negative electron gain enthalpies. Why?
Answer:
- Group 16 (chalcogens) and Group 17 (halogens) are interested to add two or one electrons respectively to attain a stable noble gas configuration.
- Because of this interest, these elements have highly negative electron gain enthalpies.
![]()
Question 16.
What is the effective nuclear charge? How is it approximated?
Answer:
The net nuclear charge experienced by valence electrons in the outermost shell is called the effective nuclear charge. It is approximated by the below-mentioned equation Zeff = Z – S.
where Z is the atomic number and S is the screening constant which can be calculated using Slater’s rules.
Question 17.
Elements Zn, Cd, and Hg with electronic configuration (n-1)d10 ns2 do not show most of the transition elements properties. Give reason.
Answer:
- Zn, Cd, and Hg are having completely filled d-orbitais (d10 electronic configuration).
- They do not have partially filled d-orbitais Like other transition elements. So they do not show much of the transition elements properties.
Question 18.
Define Ionisation energy.
Answer:
Ionization energy is defined as the minimum amount of energy required to remove the most loosely bound electron from the valence shell of the isolated neutral gaseous atom in its ground state.
Question 19.
Why d-block elements are called transition elements?
Answer:
d-block elements form a bridge between the chemically active metals of s-block elements and the less active elements of groups of 13th and 14th and thus take their familiar name transition elements.
Question 20.
Beryllium has higher ionization energy than Boron. Give reason.
Answer:
Beryllium has completely filled 2s orbital which is more stable than the partially filled valence shell electronic configuration of Boron (2s2 – 2p1). Hence, Beryllium has higher ionization energy than Boron.
![]()
Question 21.
Write the electronic configuration of lanthanides and actinides?
Answer:
- The electronic configuration of lanthanides is 4f1-144 5d0-11 6s2.
- The electronic configuration of actinides is 5f1-14 6d0-17s2.
Question 22.
What is the effect of shielding on ionization energy?
Answer:
As we move down a group, the number of inner-shell electrons increases which in turn increases the repulsive force exerted by them on the valence electrons, i.e., the increased shielding effect caused by the inner electrons decreases the attractive force acting the valence electron by the nucleus. Therefore, the ionization energy decreases.
Question 23.
Define electron affinity.
Answer:
Electron affinity is defined as the amount of energy released when an electron is added to the valence shell of an isolated neutral gaseous atom in its ground state to form its anion.
Question 24.
What are periodic properties? Give example.
Answer:
The term periodicity of properties indicates that the elements with similar properties reappear at certain regular intervals of atomic number in the periodic table.
Example:
- Atomic radii
- Ionization energy
- Electron affinity
- Electronegativity.
Question 25.
Why is the electron affinity of Nitrogen almost zero?
Answer:
Nitrogen has half-filled stable (1s2, 2s2, 2p3) electronic configuration. The addition of extra electrons will disturb their stable electronic configuration and it has almost zero electron affinity.
![]()
Question 26.
The cationic radius is smaller than its corresponding neutral atom. Justify this statement.
Answer:
- When a neutral atom loses one or more electrons it forms cation.
Na → Na+ + e– - The radius of this cation (rNa+)is decreased than its parent atom (rNa).
- When an atom is charged to cation, the number of nuclear charges becomes greater than the number of orbital electrons. Hence the remaining electrons are more strongly attracted by the nucleus. Hence the cationic radius is smaller than its corresponding neutral atom.
Question 27.
Define electronegativity.
Answer:
Electronegativity is defined as the relative tendency of an element present in a covalently bonded molecule, to attract the shared pair of electrons towards itself.
Question 28.
What are isoelectronic ions? Give example.
Answer:
There are some ions of different elements having the same number of electrons are called isoelectronic ions.
Example: Na+ Mg2+, Al3+, F–, O2-, N3-
Question 29.
What is the variation of electronegativity in a group?
Answer:
The electronegativity generally decreases down a group. As we move down a group, the atomic radius increases and the nuclear attractive force on the valence electron decreases. Hence, the electronegativity decreases.
Question 30.
The ionization energy of beryllium is greater than the ionization energy of boron. Why?
Answer:
Be (Z= 4) 1s2 2s2. it has completely filled valence electrons, which requires high IE1.
B (Z =5) 1s2 2s2 2p1. It has incompletely filled valence electrons, which requires comparatively
less IE1 Hence I.E1 Be > I.E1 B.
![]()
Question 31.
What is the diagonal relationship?
Answer:
On moving diagonally across the periodic table, the second and third-period elements show certain similarities. The similarity in properties existing between the diagonally placed elements is called the ‘diagonal relationship’.
Question 32.
Define electron gain enthalpy or electron affinity. Give its unit.
Answer:
The electron gain enthalpy of an element is the amount of energy released when an electron is added to the neutral gaseous atom.
A + electron → A– + energy (E.A)
Unit of electron affinity is KJ mole–.
![]()
II. Short Question and Answers (3 Marks):
Question 1.
How Moseley determined the atomic number of an element using X-rays?
Answer:
- Henry Moseley studied the X-ray spectra of several elements and determined their atomic numbers (Z).
- He discovered a correlation between atomic number and the frequency of X-rays generated by bombarding a clement with the high energy of electrons.
- Moseley correlated the frequency of the X-ray emitted by an equation as,
\(\sqrt{v}\) = a (Z – b)
Where υ = Frequency of the X-rays emitted by the elements.
a and b = Constants. - From the square root of the measured frequency of the X-rays emitted, he determined the atomic number of the element.
Question 2.
Write notes on Newlands classification of elements.
Answer:
Newland made an attempt to classify the elements and proposed the law of octaves. On arranging the elements in the increasing order of atomic weights, he observed that the properties of every eighth element are similar to the properties of the first element. This law holds good for lighter elements up to calcium.
Question 3.
Describe Mendeleev’s periodic classification of elements.
Answer:
Dmitriev Mendeleev proposed that “the properties of the elements are the periodic functions of their atomic weights” and this is called periodic law. Mendeleev listed the 70 known elements at that time in several vertical columns in order of increasing atomic weights. Thus, Mendeleev constructed the first periodic table based on the periodic law.
In the periodic table, he left some blank spaces since there were no known elements with the appropriate properties at that time. He and others predicted the physical and chemical properties of the missing elements. Eventually, these missing elements were discovered and found to have the predicted properties. For example, Gallium of group-III and Germanium of group- IV were unknown at that time. But Mendeleev predicted their existence and properties. After the discovery of the actual elements, their properties were found to match closely to their predicted by Mendeleev.
![]()
Question 4.
Write notes on Moseley’s work.
Answer:
Henry Mosley studied the characteristic X-rays spectra of several elements by bombarding them with high energy electrons and observed a linear correlation between atomic number and the frequency of X-rays emitted which is given by the following expression, √υ = a(Z – b)
where, υ is the frequency of the X-rays emitted by the element with atomic number ‘Z’; ‘a’ and ‘b’ are constants and have same values for all the elements. The plot of √υ against Z gives a straight line. Using this relationship, we can determine the atomic number of an unknown element from the frequency of X-rays emitted.
Question 5.
What are the reasons behind Moseley’s attempt in finding an atomic number?
Answer:
- The number of electrons increases by the same number as the increase in the atomic number.
- As the number of electrons increases, the electronic structure of the atom changes.
- Electrons in the out can not shell of an atom (valence shell electrons) determine the chemical properties of the elements.
Question 6.
What are s-block elements? Give their properties.
Answer:
The elements of group-1 and group-2 are called s-block elements, since the last valence electron enters the ns orbital.
- The group-1 elements are called alkali metals while group-2 elements are called alkaline earth metals.
- These are soft metals and possess low melting and boiling points with low ionization enthalpies.
- They are highly reactive and form ionic compounds.
- They are highly electropositive in nature and most of the elements imparts colour to the flame.
Question 7.
Explain the classification of elements based on electronic configuration.
Answer:
- The distribution of electrons into orbitais, s, p, d, and f of an atom is called its electronic configuration. The electronic configuration of an atom is characterized by a set of four quantum numbers, n, l, m, and s. of these the principal quantum number (n) defines the main energy level known as shells.
- The position of an element in the periodic table is related to the configuration of that element and thus reflects the quantum numbers of the last orbital filled.
- The electronic configuration of elements in the periodic table can be studied along with the periods and groups separately for the best classification of elements.
- Elements placed in a horizontal row of a periodic table is called a period. There are seven periods.
- A vertical column of the periodic table is called a group. A group consists of a series of elements having a similar configuration to the outermost shell. There are 18 groups in the periodic table.
![]()
Question 8.
What are d-block elements? Give their properties.
Answer:
The elements of the groups 3 to 12 are called d-block elements or transition elements with general valence shell electronic configuration ns1 – 2, (n – 1 )d1 – 10.
- These elements show more than one oxidation state and form ionic, covalent, and coordination compounds.
- They can form interstitial compounds and alloys which can also act as catalysts.
- These elements have high melting points and are good conductors of heat and electricity.
Question 9.
What is a covalent radius? Explain.
Answer:
Covalent radius is one-half of the internuclear distance between two identical atoms linked together by a single covalent bond. The internuclear distance can be determined using X-ray diffraction studies.
For example, the experimental intemuclear distance in Cl2 molecule is 1.98 Å. The covalent radius of chlorine is calculated as follows,
dcl – cl = rcl + rcl
rcl = \(\frac{d_{c l-d t}}{2}=\frac{1.98}{2}\) = 0.99 Å .
The formation of a covalent bond involves the overlapping of atomic orbitals and it reduces the expected internuclear distance. Therefore, the covalent radius is always shorter than the actual atomic radius.
Question 10.
How is the covalent radius of an individual atom can be calculated?
Answer:
The covalent radius of an individual atom can be calculated using the internuclear distance (dA-B) between two different atoms A and B. The simplest method proposed by Schomaker and Stevenson is as follows.
dA – B = rA + rB – 0.09(χA – χB)
where χA and χB are the electronegativities of A and B respectively in Pauling units. Here, χA > χB and radius is in Å.
Question 11.
Write about the electronic configuration of the 1st and 2nd periods.
Answer:
Electronic configuration of t period:
In 1 period only two elements are present. This period starts with the filling of electrons in the first energy level, n1. This level has only one orbital as is. Therefore it can accommodate two electrons maximum.
Electronic configuration of 2nd period:
In the 2’ period 8 elements are present. This period starts with the filling of electrons in the second energy level, n = 2. In this level four orbitais (one 2s and three 2p) are present. Hence the second energy level can accommodate 8 electrons. Thus, the second period has eight elements.
![]()
Question 12.
Discuss the variation of electron affinity in the period.
Answer:
As we move from alkali metals to halogens in a period, generally electron affinity increases, i.e., the amount of energy released will be more. This is due to an increase in the nuclear charge and a decrease in the size of the atoms. However, in the case of elements such as beryllium and nitrogen the addition of extra electrons will disturb their stable electronic configuration and they have almost zero electron affinity.
Noble gases have stable ns2, np6 configurations, and the addition of further electrons is unfavourable and requires energy. Halogens having the general electronic configuration of ns2, np5 readily accept an electron to get the stable noble gas electronic configuration (ns2, np6), and therefore, in each period the halogen has high electron affinity.
Question 13.
What are the two exceptions of block division in the periodic table?
Answer:
1. Helium has two electrons. Its electronic configuration is 1s2. As per the configuration, it is supposed to be placed in ‘s’ block but actually placed in s group which belongs to ‘p’ block. Because it has a completely filled valence shell as the other elements present in the 18th group. It also resembles 18th group elements in other properties. Hence helium is placed with other noble gases.
2. The other exception is hydrogen. it has only one s-electron and hence can be placed in group 1. It can also gain an electron to achieve a noble gas arrangement and hence it can behave as halogens (17th group elements). Because of these assumptions, the position of hydrogen becomes a special case. Finally, it is placed separately at the top of the periodic table.
![]()
III. Long Question and Answers (5 Marks):
Question 1.
Describe Mosley’s work and Modern Periodic Law.
Answer:
Henry Mosley studied the characteristic X-rays spectra of several elements by bombarding them with high energy electrons and observed a linear correlation between atomic number and the frequency of X-rays emitted which is given by the following expression,
√υ = a(Z – b)
where, υ is the frequency of the X-rays emitted by the element with atomic number ‘Z’; ‘a’ and ‘b’ are constants and have the same values for all the elements. The plot of √υ against Z gives a straight line. Using this relationship, we can determine the atomic number of an unknown element from the frequency of X-rays emitted.
Based on his work, the modern periodic law was developed which states that, “the physical and chemical properties of the elements are periodic functions of their atomic numbers”. Based on this law, the elements were arranged in order of their increasing atomic numbers. This mode of arrangement reveals an important truth that the elements with similar properties recur after regular intervals. The repetition of physical and chemical properties at regular intervals is called periodicity.
Question 2.
The first ionization enthalpy of magnesium is higher than that of sodium. On the other hand, the second ionization enthalpy of sodium is very much higher than that of magnesium. Explain.
Answer:
The 1st ionization enthalpy of magnesium is higher than that of Na+ due to higher nuclear charge and slightly smaller atomic radius of Mg than Na. After the loss of the first electron, N& formed has the electronic configuration of neon (2, 8). The higher stability of the completely filled noble gas configuration leads to very high second ionization enthalpy for sodium. On the other hand. Mg+ formed after losing first electron still has one more electron in its outermost (3s) orbital. As a result, the second ionization enthalpy of magnesium is much smaller than that of sodium.
Question 3.
By using Pauling’s method calculate the ionic radii of Na+ and F– ions in the potassium chloride crystal. Given that dNa+ – F– = 231 pm.
Answer:
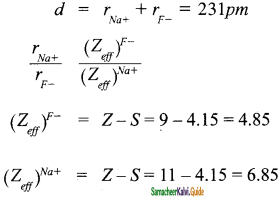
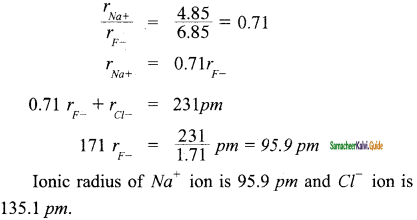
![]()
Question 4.
Discuss the variation of electronic configuration along the periods.
Answer:
Each period starts with the element having general outer electronic configuration, ns1 and ends with ns2, np6 where n is the period number. The first period starts with the filling of valence electrons in is orbital, which can accommodate only two electrons. Hence, the first period has two elements, namely hydrogen and helium.
The second period starts with the filling of valence electrons in 2s orbital followed by three 2p orbitals with eight elements from lithium to neon. The third period starts with filling of valence electrons in the 3s orbital followed by 3p orbitals. The fourth period starts with filling of valence electrons from 4s orbital followed by 3d and 4p orbitals in accordance with Aufbau principle. Similarly, we can explain the electronic configuration of elements in the subsequent periods.
In the fourth period, the filling of 3d orbitals starts with scandium and ends with zinc. These 10 elements are called first transition series. Similarly, 4d, 5d and 6d orbitals are filled in successive periods and the corresponding series of elements are called second third and fourth transition series respectively.
In the sixth period the filling of valence electrons starts with 6s orbital followed by 4f, 5d and 6p orbitals. The filling up of 4f orbitals begins with Cerium (Z = 58) and ends at Lutetium (Z = 71). These 14 elements constitute the first inner-transition series called Lanthanides. Similarly, in the seventh period 5f orbitals are filled, and it’s -14 elements constitute the second inner-transition series called Actinides. These two series are placed separately at the bottom of the modem periodic table.
Question 5.
Describe the nomenclature of elements with Atomic Number greater than 100.
Answer:
Usually, when a new element is discovered, the discoverer suggests a name following IUPAC guidelines which will be approved after a public opinion. In the meantime, the new element will be called by a temporary name coined using the following IUPAC rules until the IUPAC recognizes the new name.
1. The name was derived directly from the atomic number of the new element using the following numerical
roots.

2. The numerical roots corresponding to the atomic number are put together and ‘ium’ is added as suffix.
3. The final ‘n’ of ‘enn is omitted when it is written before ‘nil’ similarly the final ‘i’ of ‘bi’ and ‘tri’ is omitted when it written before ‘ium’.
4. The symbol of the new element is derived from the first letter of the numerical roots.

![]()
Question 6.
Explain the merits of Moseley’s long form of the periodic table.
Answer:
Merits of Moseley’s long form of the periodic table:
- As this classification is based on atomic number, it relates the position of an element to its electronic configuration.
- The elements having similar electronic configuration fall in a group. They also have similar physical and chemical properties.
- The completion of each periõd is more logical. In a period as the atomic number increases, the energy shells are gradually filled up until an inert gas configuration is reached.
- The position of zero group is also justified in the table as group 18.
- The table completely separates metals and non-metals.
- The table separates two subgroups. lanthanides and actinides, dissimilar elements do not fall together.
- The greatest advantage of this periodic table is that this can be divided into four blocks namely s, p. d, and f-block elements.
- This arrangement of elements is easier to remember, understand and reproduce.
Question 7.
Explain the variation of the atomic radius in periods and groups.
Answer:
Variation in a period:
Atomic radius tends to decrease in a period. As we move from left to right along a period, the valence electrons are added to the same shell. The simultaneous addition of protons to the nucleus increases the nuclear charge, as well as the electrostatic attractive force between the valence electrons and the nucleus. Therefore atomic radius decreases along a period.
Variation in a group:
In the periodic table, the atomic radius of elements increases down the group. As we move down a group, new shells are opened to accommodate the newly added valence electrons. As a result, the distance between the centre of the nucleus and the outermost shell containing the valence electron increases. Hence, the atomic radius increases.
Question 8.
What is an Effective nuclear charge? How is it calculated using slater’s rule?
Answer:
In addition to the electrostatic forces of attraction j between the nucleus and the electrons, there | exists repulsive forces among the electrons. The repulsive force between the inner shell electrons j and the valence electrons leads to a decrease in the j electrostatic attractive forces acting on the valence 1 electrons by the nucleus. Thus, the inner shell | electrons act as a shield between the nucleus and the valence electrons. This effect is called the shielding effect.
The net nuclear charge experienced by valence electrons in the outermost shell is called the effective 1 nuclear charge. It is approximated by the below-mentioned equation.
Zeff = Z – S
Where Z is the atomic number and ‘S’ is the screening constant which can be calculated using Slater’s rules as described below.
Step 1:
Write the electronic configuration of the atom and rearrange it by grouping ns and np orbitals together and others separately in the following form.
(1s)(2s, 2p) (3s, 3p) (3d) (4s, 4p) (4d) (4f) (5s, 5p) ……
Step 2:
Identify the group in which the electron of interest is present. The electron present right to this ! group does not contribute to the shielding effect. Each of the electrons within the identified group (denoted by ‘n’) shields to, an extent of 0.35 unit of nuclear charge. However, it is 0.30 unit for 1s electron.
Step 3:
Shielding of inner-shell electrons. If the electron of interest belongs to either s or p orbital,
(i) each electron within the (n – 1) group shields to an extent of 0.85 unit of nuclear charge, and
(ii) each electron within the (n – 2) group (or) even lesser group (n – 3), (n – 4) etc…
completely shields i.e. to an extent of 1.00 unit of nuclear charge.
If the electron of interest belongs to d or f orbital, then each of electron left of the group of electron of interest shields to an extent of 1.00 unit of nuclear charge.
Step 4:
Summation of the shielding effect of all the electrons gives the shielding constant ‘S’.
![]()
Question 9.
Explain the periodic variation of ionization energy in a period.
Answer:
The ionization energy usually increases along a period with few exceptions. When we move from left to right along a period, the valence electrons are added to the same shell, at the same time protons are added to the nucleus, This successive increase of nuclear charge increases the electrostatic attractive force on the valence electron and more energy is required to remove the valence electron resulting in high ionization energy. Consider the variation in ionization energy of second period elements. The plot of atomic number vs ionisation energy is given below.
In the graph, there are two deviations in the trends of ionisation energy. It is expected that boron has higher ionisation energy than beryllium since it has higher nuclear charge. However, the actual ionisation energies of beryllium and boron are 899 and 800kJ mol-1 respectively contrary to the expectation. It is due to the fact that beryllium with completely filled 2s orbital, is more stable than partially filled valence shell electronic configuration of boron. (2s2, 2p1).
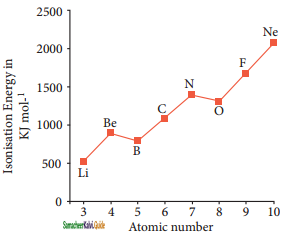
The electronic configuration of beryllium (Z = 4) in its ground state is 1s2, 2s2 and that of boron (Z = 5) 1s2, 2s2, 2p1. Similarly, nitrogen with 1s2, 2s2, 2p1 electronic configuration has higher ionisation energy (l402 kJmol-1) than oxygen (1314 kJmol-1). Since the half-filled electronic configuration is more stable, it requires higher energy to remove an electron from 2p orbital of nitrogen. Whereas the removal of one 2p electron from oxygen leads to a stable half-filled configuration. This makes it comparatively easier to remove 2p electron from oxygen.
Question 10.
Explain the periodic variation of electron affinity in a group.
Answer:
Electron affinity is defined as the amount of energy released (required in the case of noble gases) when an electron is added to the valence shell of an isolated ^neutral gaseous atom in its ground state to form its anion. It is expressed in kJmol-1
A + 1e– → A– + EA
Variation of Electron Affinity in a period:
As we move from alkali metals to halogens in a period, generally electron affinity increases, i.e., the amount of energy released will be more. This is due to an increase in the nuclear charge and a decrease in size of the atoms. However, in case of elements such as beryllium (1s2, 2s2), nitrogen (1s2, 2s2, 2p3) the addition of extra electron will disturb their stable electronic configuration and they have almost zero electron affinity.
Noble gases have stable ns2, np6 configurations, and the addition of further electrons is unfavourable and requires energy. Halogens having the general electronic configuration of ns2, np5 readily accept an electron to get the stable noble gas electronic configuration (ns2, np6), and therefore in each period the halogen has high electron affinity, (high negative values)
Question 11.
Define electron affinity. How does it vary along with the group?
Answer:
Electron affinity is defined as the amount of energy released (required in the case of noble gases) when an electron is added to the valence shell of an isolated neutral gaseous atom in its ground state to form its anion. It is expressed in kJmoF1
A + 1e– → A– + EA
Variation of Electron affinity in a group:
As we move down a group, generally the electron affinity decreases. It is due to an increase in atomic size and the shielding effect of inner-shell electrons. However, oxygen and fluorine have lower affinity than sulphur and chlorine respectively. The sizes of oxygen and fluorine atoms are comparatively, small and they have high electron density.
Moreover, the extra electron, added to oxygen and fluorine has to be accommodated in the 2p orbital which is relatively compact compared to the 3p orbital of sulphur and chlorine so, oxygen and fluorine have lower electron affinity than their respective group elements sulphur and chlorine.
![]()
Question 12.
Explain the salient features of groups.
Answer:
1. Number of electrons in the outermost shell:
The number of electrons present in the outermost shells does not change on moving down in a group, i.e remains the same. Hence, the valency also remains the same within a group.
2. Number of shells:
In going down a group the number of shells increases by one at each step and ultimately becomes equal to the period number to which the element belongs.
3. Valency:
The valencies of all the elements of the same group are the same. The valency of an element with respect to oxygen is same in a group.
4. Metallic character:
The metallic character of the elements increases in moving from top to bottom in a group.