Tamilnadu State Board New Syllabus Samacheer Kalvi 11th Chemistry Guide Pdf Chapter 8 Physical and Chemical Equilibrium Text Book Back Questions and Answers, Notes.
Tamilnadu Samacheer Kalvi 11th Chemistry Solutions Chapter 8 Physical and Chemical Equilibrium
11th Chemistry Guide Physical and Chemical Equilibrium Text Book Back Questions and Answers
Textbook Evaluation:
I. Choose the best answer:
Question 1.
If Kb and Kf for a reversible reaction are 0.8 × 10-5 and 1.6 × 10-4 respectively, the value of the equilibrium constant is,
a) 20
b) 0.2 × 10-1
c) 0.05
d) None of these
Answer:
a) 20
Question 2.
At a given temperature and pressure, the equilibrium constant values for the equilibria
3A2 + B2 + 2Cr ![]() 2A3BC and
2A3BC and
A3BC ![]() \(\frac{3}{2}\) [A2] + \(\frac{1}{2}\) B2 +C
\(\frac{3}{2}\) [A2] + \(\frac{1}{2}\) B2 +C
The relation between K1 and K2 is
a) K1 = \(\frac{1}{\sqrt{\mathrm{K}_{2}}}\)
b)K2 = \(\mathrm{K}_{1}^{-1 / 2}\)
C) K12 = 2K2
d) \(\frac{\mathrm{K}_{1}}{2}\) = K2
Answer:
b)K2 = \(\mathrm{K}_{1}^{-1 / 2}\)
Question 3.
The equilibrium constant for a reaction at room temperature is K1 and that at 700 K is K2 If K1 > K2, then
a) The forward reaction is exothermic
b) The forward reaction is endothermic
c) The reaction does not attain equilibrium
d) The reverse reaction is exothermic
Answer:
a) The forward reaction is exothermic
Question 4.
The formation of ammonia from N2(g) and H2(g) is a reversible reaction 2NO(g) + O2(g) ![]() 2NO2(g) + Heat What is the effect of increase of temperature on this equilibrium reaction
2NO2(g) + Heat What is the effect of increase of temperature on this equilibrium reaction
a) equilibrium is unaltered
b) formation of ammonia is favoured
c) equilibrium is shifted to the left
d) reaction rate does not change
Answer:
c) equilibrium is shifted to the left
Question 5.
Solubility of carbon dioxide gas in cold water can be increased by
a) increase in pressure
b) decrease in pressure
c) increase in volume
d) none of these
Answer:
a) increase in pressure
![]()
Question 6.
Which one of the following is incorrect statement?
a) for a system at equilibrium, Q is always less than the equilibrium constant
b) equilibrium can be attained from either side of the reaction
c) the presence of catalyst affects both the forward reaction and reverse reaction to the same extent
d) Equilibrium constant varied with temperature
Answer:
a) for a system at equilibrium, Q is always less than the equilibrium constant
Question 7.
K1 and K2 are the equilibrium constants for the reactions respectively.
N2(g) + O2(g)![]() 2NO(g)
2NO(g)
2NO(g) + O2(g) ![]() 2NO2 (g)
2NO2 (g)
What is the equilibrium constant for the reaction NO2(g) ⇌ 1/2 N2(g) + O2(g)
a) \(\frac{1}{\sqrt{\mathrm{K}_{1} \mathrm{~K}_{2}}}\)
b) \(\left(\mathrm{K}_{1}=\mathrm{K}_{2}\right)^{1 / 2}\)
c) \(\frac{1}{2 \mathrm{~K}_{1} \mathrm{~K}_{2}}\)
d) \(\left(\frac{1}{\mathrm{~K}_{1} \mathrm{~K}_{2}}\right)^{3 / 2}\)
Answer:
a) \(\frac{1}{\sqrt{\mathrm{K}_{1} \mathrm{~K}_{2}}}\)
Question 8.
In the equilibrium, 2A(g) ⇌ 2B(g) + C2(g) the equilibrium concentrations of A, B and C2 at 400K are 1 × 10-4M, 2.0 × 10-3 M, 1.5 × 10-4 M respectively. The value of Kc for the equilibrium at 400 K is
a) 0.06
b) 0.09
c) 0.62
d) 3 × 10-2
Answer:
a) 0.06
Question 9.
An equilibrium constant of 3.2 × 10-6 for a reaction means, the equilibrium is
a) largely towards forward direction
b) largely towards reverse direction
c) never established
d) none of these
Answer:
b) largely towards reverse direction
Question 10.
\(\frac{\mathrm{K}_{\mathrm{C}}}{\mathrm{K}_{\mathrm{P}}}\) for the reaction,
N2(g) + 3H2(g) ⇌ 2NH3 (g) is
a) \(\frac{1}{\mathrm{RT}}\)
b) \(\sqrt{\mathrm{RT}}\)
c) RT
d) (RT)2
Answer:
d) (RT)2
![]()
Question 11.
For the reaction AB (g) ⇌ A(g) + B(g), at equilibrium, AB is 20 % dissociated at a total pressure of P, the equilibrium constant Kp is related to the total pressure by the expression
a) P = 24 Kp
b) P = 8 Kp
c) 24 P = Kp
d) none of these
Answer:
a) P = 24 Kp
Question 12.
In which of the following equilibrium, Kp and Kc are not equal?
a) 2N0(g) ⇌ N2(g) + O2(g)
b) SO2(g) + NO2 ⇌ SO3(g) + NO(g)
c) H2(g) + I2(g) ⇌ 2HI(g)
d) PCl5 ⇌ PCl3(g) + Cl2(g)
Answer:
b) SO2(g) + NO2 ⇌ SO3(g) + NO(g)
Question 13.
If x is the fraction of PCl5 dissociated at equilibrium in the reaction PCl5 ⇌ PCl3 + Cl2 then starting with 0.5 mole of PCl5, the total number of moles of reactants and products at equilibrium is
a) 0.5 – x
b) x + 0.5
c) 2x + 0.5
d) x + 1
Answer:
b) x + 0.5
Question 14.
The values of Kp1 and Kp2; for the reactions, X ⇌ Y + Z, A ⇌ 2B are in the ratio 9 : 1 if degree of dissociation of X and A be equal then total pressure at equilibrium P1, and P2 are in the ratio
a) 36 : 1
b) 1 : 1
c) 3 : 1
d) 1 : 9
Answer:
a) 36 : 1
Question 15.
In the reaction
Fe(OH)3(S) ⇌ Fe3+ (aq) + 3OH– (aq),
if the concentration of OH– ions is decreased by 1/4 times, then the equilibrium concentration of Fe3+ will
a) not changed
b) also decreased by 1/4 times
c) increase by 4 times
d) increase by 64 times
Answer:
d) increase by 64 times
![]()
Question 16.
Consider the reaction where Kp = 0.5 at a particular temperature PCl5(g) ⇌ PCl3 (g) + Cl2 (g) if the three gases are mixed in a container so that the partial pressure of each gas is initially 1 atm, then which one of the following is true.
a) more PCl3 will be produced
b) more Cl2 will be produced
c) more PCl5 will be produced
d) none of these
Answer:
c) more PCl5 will be produced
Question 17.
Equimolar concentrations of H2 and I2 are heated to equilibrium in a 1 litre flask. What percentage of the initial concentration of H2 has reacted at equilibrium if rate constant for both forward and reverse reactions are equal
a) 33%
b) 66%
c) (33)2%
d) 16.5 %
Answer:
a) 33%
Question 18.
In a chemical equilibrium, the rate constant for the forward reaction is 2.5 × 10-2, and the equilibrium constant is 50. The rate constant for the reverse reaction is,
a) 11.5
b) 50
c) 2 × 102
d) 2 × 10-3
Answer:
b) 50
Question 19.
Which of the following is not a general characteristic of equilibrium involving physical process
a) Equilibrium is possible only in a closed system at a given temperature
b) The opposing processes occur at the same rate and there is a dynamic but stable condition
c) All the physical processes stop at equilibrium
d) All measurable properties of the system remains constant
Answer:
c) All the physical processes stop at equilibrium
Question 20.
For the formation of Two moles of SO3(g) from SO2 and O2, the equilibrium constant is K1. The equilibrium constant for the dissociation of one mole of SO3 into SO2 and O2 is
a) \(1 / \mathrm{K}_{1}\)
b) K12
c) \(\left(\frac{1}{\mathrm{~K}_{1}}\right)^{1 / 2}\)
d) \(\frac{\mathrm{K}_{1}}{2}\)
Answer:
c) \(\left(\frac{1}{\mathrm{~K}_{1}}\right)^{1 / 2}\)
![]()
Question 21.
Match the equilibria with the corresponding conditions:
i) Liquid ⇌ Vapour
ii) Solid ⇌ Liquid
iii) Solid ⇌ Vapour
iv) Solute(s) ⇌ Solute (Solution)
1) Melting point
2) Saturated solution
3) Boiling point
4) Sublimation point
5) Unsaturated solution
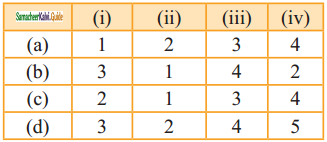
Answer:
b) 3 1 4 2
Question 22.
Consider the following reversible reaction at equilibrium, A + B ⇌ C, If the concentration of the reactants A and B are doubled, then the equilibrium constant will
a) be doubled
b) become one fourth
c) be halved
d) remain the same
Answer:
d) remain the same
Question 23.
[Co(H2O)6]2+ (aq) (pink) + 4Cl– (aq) ⇌ [CoCl4]2-(aq) (blue) + 6 H2O (l)
In the above reaction at equilibrium, the reaction mixture is blue in colour at room temperature. On cooling this mixture, it becomes pink in color. On the basis of this information, which one of the following is true?
a) ∆H > 0 for the forward reaction
b) ∆H = 0 for the reverse reaction
c) ∆H < 0 for the forward reaction
d) Sign of the ∆H cannot be predicted based on this information
Answer:
a) ∆H > 0 for the forward reaction
Question 24.
The equilibrium constants of the following reactions are:
N2 + 3H2 ⇌ 2NH3; K1
N2 + O2 ⇌ 2NO; K2
H2 + 1/2O2 ⇌ H2O; K3
The equilbrium constant (K) for the reaction ;
2NH3 + 5/2 O2 ⇌ 2NO + 3H2O, will be
a) K23 K3/K1
b) K1 K33/K2
c) K2K33/K1
d) K2K3/K1
Answer:
c) K2K33/K1
Question 25.
A 20 litre container at 400 K contains CO2 (g) at pressure 0.4 atm and an excess (neglect the volume of solid SrO). The volume of the container is now decreased by moving the movable piston fitted in the container. The maximum volume of the container, when the pressure of CO2 attains its maximum value will be: Given that: SrCO3(S) ≅ SrO + CO2 (g) [Kp = 1.6 atm]
a) 2 litre
b) 5 litre
c) 10 litre
d) 4 litre
Answer:
b) 5 litre
![]()
II. Write brief answer to the following questions:
Question 26.
If there is no change in concentration, why is the equilibrium state considered dynamic?
Answer:
Rate of forward reaction = Rate of backward reaction
Question 27.
For a given reaction at a particular temperature, the equilibrium constant has constant value. Is the value of Q also constant? Explain.
Answer:
Kc and Qc are constant at equilibrium, both are temperature dependent. When Kc is constant at given temperature, Qc also constant.
Question 28.
What is the relation between Kp and Kc? Given one example for which Kp is equal to Kc.
Answer:
Kp = Kc(RT)∆ng
H2(g) + I2(g) ≅ 2HI (g) ; ∆ng = 0 ;
Kp = Kc.
Question 29.
For a gaseous homogeneous reaction at equilibrium, number of moles of products are greater than the number of moles of reactants. Is Kc is larger or smaller than Kp.
Answer:
Kp >Kc
np > nR
Question 30.
When the numerical value of the reaction quotient (Q) is greater than the equilibrium constant, in which direction does the reaction proceed to reach equilibrium?
Answer:
Q > Kc
Reverse reaction is favoured.
![]()
Question 31.
For the reaction, A2(g) + B2(g) ⇌ 2AB(g); ∆H is -ve.
the following molecular scenes represent differenr reaction mixture.(A-green, B-blue)
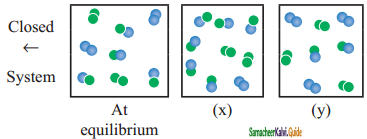
i) Calculate the equilibrium constant Kp and (Kc).
ii) For the reaction mixture represented by scene (x), (y) the reaction proceed in which directions?
iii) What is the effect of increase in pressure for the mixture at equilibrium.
Answer:
Kc = \(\frac{[\mathrm{AB}]^{2}}{\left[\mathrm{~A}_{2}\right]\left[\mathrm{B}_{2}\right]}\) ; A – green, B – blue
Given that ‘V’ is constant(closed system)
At equilbrium,
Kc = \(\frac{(-)}{\left(\frac{2}{V}\right)\left(\frac{2}{V}\right)}\) = \(\frac{16}{4}\) = 4
Kp = Kc(RT)∆n = 4 (RT)0 = 4
At stage ‘x’,
Q = \(\frac{\left(\frac{6}{\mathrm{~V}}\right)^{2}}{\left(\frac{2}{\mathrm{~V}}\right)\left(\frac{1}{\mathrm{~V}}\right)}=\frac{36}{2}\) = 18
Q > Kc i.e., reverse reaction is favoured.
At stage ‘y’,
Q = \(\frac{\left(\frac{3}{V}\right)^{2}}{\left(\frac{3}{V}\right)\left(\frac{3}{V}\right)}=\frac{9}{3 \times 3}\) = 1
Kc > Q i.e., forward reaction is favoured.
Question 32.
State Le – Chateller principle.
Answer:
“If a system at equilibrium is distributed, then the system shifts itself in a direction that nullifies the effect of that disturbance”.
Question 33.
Consider the following reactions,
a) H2(g) + I2(g) ⇌ 2HI(g)
b) CaCO3(s) ⇌ CaO (s) + CO2(g)
c) S(s) + 3 F2(g) ⇌ SF6(g)
In each of the above reactions find out whether you have to increase (or) decrease the volume to increase the yield of the product.
Answer:
a) H2(g) + I2(g) ⇌ 2HI(g)
In this reaction, there is no effect on changing the volume. ∆ng = 0
b) CaCO3(s) ⇌ CaO (s) + CO2(g)
In this reaction, increases in volume favours forward reaction.
c) S(s) + 3 F2(g) ⇌ SF6(g)
In this reaction, decreases in volume favours forward reaction.
Question 34.
State law of mass action.
Answer:
The law states that, “At any instant, the rate of a chemical reaction at a given temperature is directly proportional to the product of the active masses of the reactants at that instant”.
Rate ∝ [Reactant]x
Active mass = \(\left(\frac{\mathrm{n}}{\mathrm{V}}\right)\) mol dm-3 (or) mol L-1
![]()
Question 35.
Explain how will you predict the direction of an equilibrium reaction.
Answer:
From the knowledge of equilibrium constant, it is possible to predict the di¬rection in which the net reaction is taking place for a given concentration or partial pressure of reactants and products.
Consider a general homogeneous reversible reaction,
xA + yB ⇌ lC + mD
For the above reaction under non-equilibrium conditions, reaction quotient ‘Q’ is defined as the ratio of the product of active masses of reaction products raised to the respective stiochiometric coefficients in the balanced chemical equation to that of the reactants.
Under non – equilibrium conditions, the reaction quotient Q can be calculated using the following expression.
Q = \(\frac{[\mathrm{C}]^{1}[\mathrm{D}]^{\mathrm{m}}}{[\mathrm{A}]^{\mathrm{x}}[\mathrm{B}]^{\mathrm{y}}}\)
As the reaction proceeds, there is a continuous change in the concentration of reactants and products and also the Q value until the reaction reaches equilibrium. At equilibrium, Q is equal to Kc at a particular temperature. Once the equilibrium is attained, there is no change in the Q value. By knowing the Q value, we can predict the direction of the reaction by comparing it with Kc.
If Q = Kc, the reaction is in equilibrium state.
If Q > Kc, the reaction will proceed in the reverse direction, i.e., formation of reactants.
If Q < Kc, the reaction will proceed in the forward direction i.e., formation of products.
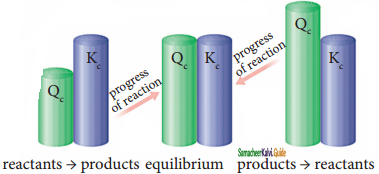
Predicting the direction of a reaction
Question 36.
Derive a general expression for the equilibrium constant Kp and Kc for the reaction, 3H2(g) + N2(g) ⇌ 2NH3(g).
Answer:
Let us consider the formation of ammonia in which, ‘a’ moles nitrogen and h’ moles hydrogen gas are allowed to react in a container of volume V. Let ‘x’ moles of nitrogen react with 3 x moles of hydrogen to give 2x moles of ammonia.
3H2(g) + N2(g) ⇌ 2NH3(g)
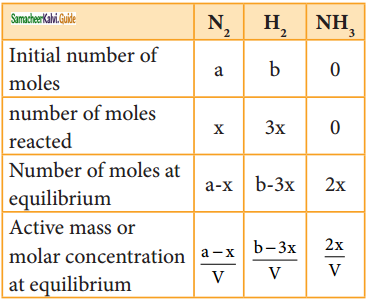
Applying law of mass action,
K2 = \(\frac{\left[\mathrm{NH}_{3}\right]^{2}}{\left[\mathrm{~N}_{2}\right]\left[\mathrm{H}_{2}\right]^{3}}\)

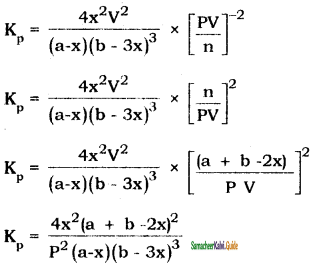
Question 37.
Write the balanced chemical equation for an equilibrium reaction for which the equilibrium constant is given by expression.
Answer:
Kc = \(\frac{\left[\mathrm{NH}_{3}\right]^{4}\left[\mathrm{O}_{2}\right]^{7}}{\left.[\mathrm{NO}]^{4} \mid \mathrm{H}_{2} \mathrm{O}\right]^{6}}\)
Balanced equation is:
4NO + 6H2O ⇌ 4NH3 + 7O2
Question 38.
What is the effect of added Inert gas on the reaction at equilibrium?
Answer:
When an inert gas (i.e., a gas which does not react with any other species involved in equilibrium) is added to an equilibrium system at constant volume, the total number of moles of gases present in the container increases, that is, the total pressure of gases increases. The partial pressure of the reactants and the products are unchanged. Hence at constant volume, the addition of inert gas has no effect on the equilibrium.
![]()
Question 39.
Derive the relation between Kp and Kc.
Answer:
Let us consider the general reaction in which all reactants and products are ideal gases.
xA + yB ⇌ lC + mD
The equilibrium constant, Kc is,
Kc = \(\frac{\left[\mathrm{Cl}^{l}[\mathrm{D}]^{\mathrm{m}}\right.}{[\mathrm{A}]^{\mathrm{x}}[\mathrm{B}]^{\mathrm{y}}}\) ……………..(1)
and Kp is,
Kp = \(\frac{p_{C}^{l} \times p_{D}^{m}}{p_{A}^{x} \times p_{B}^{y}}\) ………….(2)
The ideal gas equation is,
PV = nRT
P = \(\frac{n}{V}\) RT
since, Active mass = molar concentration = n/V
P = active mass × RT
Based on the above expression the partial pressure of the reactants and products can be expresssed as,
PXA = [A]x[RT]x
PYB = [A]y[RT]y
PlC = [A]l[RT]l
PmD = [A]m[RT]m
On substituting in Eqn.(2).,

By comparing equation (1) and (4),
we get
Kp∆ng =Kc (RT) …………..(5)
Where,
∆ng is the difference between the sum of number of moles of products and the sum of number of moles of reactants in the gas phase.
The following relations become immediately obvious.
when ∆ng = 0.
Kp = Kc (RT)0 = Kc
Example :
H2(g) + I2(g) ⇌ 2HI(g)
N2 + O2 ⇌ 2NO(g)
when ∆ng = +ve
Kp = Kc(RT)+ve
Kp > Kc
Example:
2NH3(g) ⇌ N2(g) + 3H2(g)
PCl5(g) ⇌ PCl3(g) + Cl2(g)
When ∆ng = -ve
Kp = Kc(RT)-ve
Kp < Kc
Example:
2H2(g) + O2(g) ⇌ 2H2O(g)
2SO2(g)+ O2(g) ⇌ 2SO3(g)
Question 40.
One mole of PCl5 is heated in one litre closed container. If 0.6 mole of chlorine is found at equilibrium, Calculate the value of equilibrium constant.
Solution:
Given that,
[PCl5]initial = \(\frac{1 \text { mole }}{1 \mathrm{dm}^{3}}\)
[Cl2]eq = 0.6 mole dm-3
PCl5 ⇌ PCl3 + Cl2
[PCl3]eq = 0.6 mole dm-3
[PCl5]eq = 0.4 mole dm-3
∴ Kc = \(\frac{\left[\mathrm{PCl}_{3}\right]\left[\mathrm{Cl}_{2}\right]}{\left[\mathrm{PCl}_{5}\right]}=\frac{0.6 \times 0.6}{0.4}\)
= 0.9 mole dm-3
Question 41.
For the reaction SrCO3 (s) ⇌ SrO(s) + CO2(g) the value of equilibrium constant Kp = 2.2 × 10-4 at 1002 K. Calculate Kc for the reaction.
Solution :
For the reaction,
SrCO3(s) ⇌ SrO(s) + CO2(g)
∆ng = 1 – 0 = 1
∴ Kp = Kc (RT)
2.2 × 10-4 = Kc (0.0821) (1002)
Kc = \(\frac{2.2 \times 10^{-4}}{0.0821 \times 1002}\) = 2.674 × 10-6
Question 42.
To study the decomposition of hydrogen iodide, a student fills an evacuated 3 litre flask with 0.3 mol of HI gas and allows the reaction to proceed at 500°C. At equilibrium he found the concentration of HI which is equal to 0.05 M. Calculate Kc and Kp.
Solution :
V = 3 L
[HI]initial = \(\frac{0.3 \mathrm{~mol}}{3 \mathrm{~L}}\) 0.1 M
[HI]eq = 0.05 M
2 HI(g) ⇌ H2(g) + I2(g)
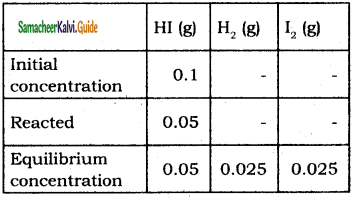
Kc = \(\frac{\left[\mathrm{H}_{2}\right]\left[\mathrm{I}_{2}\right]}{[\mathrm{HI}]^{2}}=\frac{0.025 \times 0.025}{0.05 \times 0.05}\) = 0.25
Kp = Kc(RT)(∆ng)
∆ng = 2 – 2 = 0
Kp = 0.25(RT)0
Kp = 0.25.
![]()
Question 43.
Oxidation of nitrogen monoxide was studied at 200 with initial pressures of 1 atm NO and 1 atm of O2. At equilibrium partial pressure of oxygen is found to be 0. 5 atm calculate Kp value.
Solution:
2 NO(g) + O2(g) ⇌ 2NO2(g)
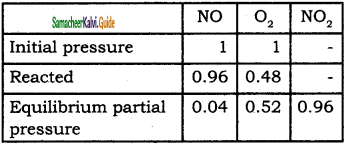
Kp = \(\frac{\left(P_{N O_{2}}\right)^{2}}{\left(P_{N O}\right)^{2}\left(P_{O}\right)}\)
= \(\frac{0.96 \times 0.96}{0.04 \times 0.04 \times 0.52}\)
Kp = 1.017 × 103.
Question 44.
1 mol of CH4, 1 mole of CS2 and 2 mol of H2S are 2 mol of H2 are mixed in a 500 ml flask. The equilibrium constant for the reaction Kc = 4 x 10-2 mol2 lit-2. In which direction will the reaction proceed to reach equilibrium ?
Solution :
CH4(g) + 2 H2S (g) ⇌ CS2(g) + 4H2(g)
Kc = 4 x 10-2 mol2 lit-2
Volume = 500 ml = \(\frac{1}{2}\) L
[CH4]in = \(\frac{1 \mathrm{~mol}}{\frac{1}{2} L}\) = 2 mol L-1;
[CH2]in = \(\frac{1 \mathrm{~mol}}{\frac{1}{2} L}\) = 2 mol L-1
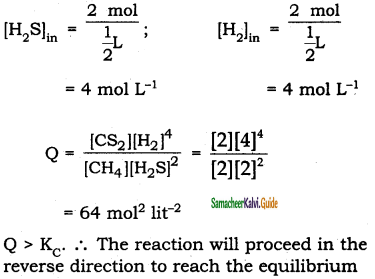
Question 45.
At particular temperature Kc = 4 × 10-2 for the reaction, H2S ⇌ 2H2(g) + S2(g). Calculate the Kc for each of the following reaction.
i) 2H2S(g) ⇌ 2H2 + S2(g)
ii) 3H2S(g) ⇌ 3H2(g) + 3/2 S2(g)
Solution :
Kc = 4 × 10-2 for the reaction,
H2S ⇌ 2H2(g) + S2(g)
Kc = \(\frac{\left[\mathrm{H}_{2}\right]\left[\mathrm{S}_{2}\right]^{1 / 2}}{\left[\mathrm{H}_{2} \mathrm{~S}\right]}\)
⇒ 4 × 10-2 = \(\frac{\left[\mathrm{H}_{2}\right]\left[\mathrm{S}_{2}\right]^{1 / 2}}{\left[\mathrm{H}_{2} \mathrm{~S}\right]}\)
(i) 2H2S(g) ⇌ 2H2 + S2(g)
For the reaction,
2H2S(g) ⇌ 2H2 + S2(g)
Kc = \(\frac{\left[\mathrm{H}_{2}\right]^{2}\left[\mathrm{~S}_{2}\right]}{\left[\mathrm{H}_{2} \mathrm{~S}\right]^{2}}\)
= (4 × 10-2)2 = 16 × 10-4
(ii) 3H2S(g) ⇌ 3H2(g) + 3/2 S2(g)
For the reaction,
3H2S(g) ⇌ 3H2(g) + 3/2 S2(g)
Kc = \(\frac{\left[\mathrm{H}_{2}\right]^{3}\left[\mathrm{~S}_{2}\right]^{3 / 2}}{\left[\mathrm{H}_{2} \mathrm{~S}\right]^{3}}\)
= (4 × 10-2)3 = 64 × 10-6
![]()
Question 46.
28 g of Nitrogen and 6 g of hydrogen were mIed In a 1 litre closed container. At equIlibrium 17 g NH3 was produced. Calculate the weight of nitrogen, hydrogen at equilibrium.
Solution:
Given mN2 =28g;
mH2 = 6g;
V = 1 L.
(nN2)initial = \(\frac{28}{28}\) = 1
(nH2)initial = \(\frac{6}{2}\) = 3
N2(g) + 3 H2(g) ⇌ 2NH3(g)
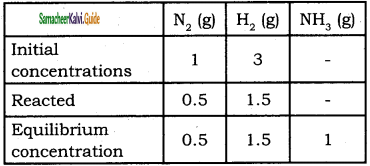
[NH3] = \(\frac{17}{17}\) = 1 mol
Weight of N2 = (no. of moles of N2) × (molar mass of N2)
= 0.5 × 28 = 14g
Weight of H2 = (no. of moles of H2) × (molar mass of H2)
= 1.5 × 2 = 3g
Question 47.
The equilibrium for the dissociation of XY2 is given as,
2 XY2(g) ⇌ 2 XY(g) + Y2(g)
if the degree of dissociation x is so small compared to one. Show that 2 Kp = PX3 where P is the total pressure and Kp is the dissociation equilibrium constant of XY2.
Answer:
2 XY2(g) ⇌ 2 XY(g) + Y2(g)

Question 48.
A sealed container was filled with 0.3 mol H2(g), 0.4 mol I2(g) and 0.2 mol HI(g) at 800 K and total pressure 1.00 bar. Calculate the amounts of the components in the mixture at equilibrium given that K = 870 for the reaction, H2(g) + I2(g) ⇌ 2 HI (g).
Solution:
A2(g) + B2(g) ⇌ 2 AB (g)

Given that, Kp = 1;
\(\frac{4 x^{2}}{(1-x)^{2}}\) = 1
⇒ 4x2 = (1 – x)2 = 1
⇒ 4x2 = 1 + x2 – 2x
3x2 + 2x – 1 = 0
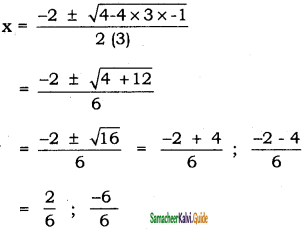
x = 0.33 – 1(not possible)
∴ [A2]eq = 1 – x = 1 – 0.33 = 0.67
[B2]eq = 1 – x = 1 – 0.33 = 0.67
[AB2]eq = 2x × 0.33 = 0.66
Question 49.
Deduce the Vant Hoff equation.
Answer:
This equation gives that quantitative temperature dependence of equilibrium constant (K). The relation between standard free energy change (∆G°) and equilibrium constant is
∆G° = – RT In K ………………..(1)
We know that, ∆G° = ∆H° – T∆S° …………(2)
Substituting (1) in equation (2)
– RT In K = ∆H° – T∆S°
Rearranging, In K = \(\frac{\Delta \mathrm{H}^{0}}{\mathrm{RT}}+\frac{\Delta \mathrm{S}^{0}}{\mathrm{RT}}\) ……………(3)
Differentiating equation (3) with respect to temperature,
\(\frac{\mathrm{d}(\ln \mathrm{K})}{\mathrm{dT}}=\frac{\Delta \mathrm{H}^{0}}{\mathrm{RT}^{2}}\) ……………….(4)
Equation (4) is known as differential form of van,t Hoff equation.
On integrating the equation (4), between T1 and T2 with their respective equilbrium consatnts K1 and K2.
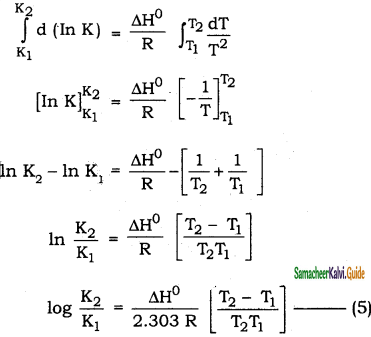
Equation 5 is known as integrated form of Van’t Hoff equation.
Question 50.
The equilibrium constant Kp for the reaction N2(g) + 3H2(g) ⇌ 2NH3(g) is 8.19 × 102 at 298 K and 4.6 × 10-1 at 498 K. Calculate ∆H° for the reaction.
Solution :
Kp1 = 8.19 × 102;
T1 = 298 K
Kp1 = 8.19 × 102;
T1 = 298 K
Kp2 = 4.16 × 10-1;
T2 = 498 K

Question 51.
The partial pressure of carbon dioxide in the reaction CaCO3(s) ⇌ CaO(s) + CO2(g) is 1.017 × 10-3 atm at 500°C. Calculate Kp at 600°C for the reaction. H for the reaction is 181 KJ mol-1 and does not change in the given range of temperature.
Solution:
PCO2 = 1.017 × 10-3 atm
T = 500°C;
Kp = PCO2
∴ Kp1 = 1.017 × 10-3;
T = 500 + 273 = 773 K
Kp2 = ?
T = 600 + 273 = 873 K
∆H° = 181 KJ mol-1

![]()
11th Chemistry Guide Physical and Chemical Equilibrium Additional Questions and Answers
I.Choose the best answer:
Question 1.
For which of the following Kp is less than Kc?
a) N2O4 ⇌ 2NO2
b) N2 + 3H2 ⇌ 2NH3
c) H2 + I2 ⇌ 2HI
d) CO + H2O ⇌ CO2 + H2
Answer:
b) N2 + 3H2 ⇌ 2NH3
Question 2.
In which of the following reaction, the value of Kp will be equal to Kc?
a) H2 + I2 ⇌ 2HI
b) PCl5 ⇌ PCl3 + Cl2
c) 2NH3 ⇌ N2 + 3H2
d) 2SO2 +O2 ⇌ 2SO3
Answer:
a) H2 + I2 ⇌ 2HI
Question 3.
For homogeneous gas reaction 4NH3 + 5O2 ⇌ 4NO + 6H2O. The equilibrium constant Kc has the unit
a) (concentration)1
b) (concentration)1
c) (concentration)9
d) (concentration)10
Answer:
a) (concentration)1
Question 4.
The reaction, 2SO2(g) + O2(g) ⇌ 2SO3(g) is carried out in a 1 dm3 vessel and 2 dm3 vessel separately. The ratio of the reaction velocities will be
a) 1 : 8
b) 1 : 4
c) 4 : 1
d) 8 : 1
Answer:
d) 8 : 1
Question 5.
Kp for the following reaction at 700 K is 1.3 × 10-3 atm-1. The Kc same temperature for the reaction. 2SO2 + O2 ⇌ 2SO3 will be
a) 1.1 × 10-2
b) 3.1 × 10-2
c) 5.2 × 10-2
d) 7.4 × 10-2
Answer:
d) 7.4 × 10-2
![]()
Question 6.
The equilibrium constant expression for the equilibrium :
2NH2(g) + 2O2(g) ⇌ N2O(g) + 3H2O(g) is
a) Kc = \(\frac{\left[\mathrm{N}_{2} \mathrm{O}\right]\left[\mathrm{H}_{2} \mathrm{O}\right]^{3}}{\left[\mathrm{NH}_{3}\right]\left[\mathrm{O}_{2}\right]}\)
b) Kc = \(\frac{\left[\mathrm{H}_{2} \mathrm{O}\right]^{3}\left[\mathrm{~N}_{2} \mathrm{O}\right]}{\left[\mathrm{NH}_{3}\right]^{2}\left[\mathrm{O}_{2}\right]^{2}}\)
c) Kc = \(\frac{\left[\mathrm{NH}_{3}\right]^{2}\left[\mathrm{O}_{2}\right]^{2}}{\left[\mathrm{~N}_{2} \mathrm{O}\right]\left[\mathrm{H}_{2} \mathrm{O}\right]^{3}}\)
d) Kc = \(\frac{\left[\mathrm{NH}_{3}\right]\left[\mathrm{O}_{2}\right]}{\left[\mathrm{N}_{2} \mathrm{O}\right]\left[\mathrm{H}_{2} \mathrm{O}\right]}\)
Answer:
b) Kc = \(\frac{\left[\mathrm{H}_{2} \mathrm{O}\right]^{3}\left[\mathrm{~N}_{2} \mathrm{O}\right]}{\left[\mathrm{NH}_{3}\right]^{2}\left[\mathrm{O}_{2}\right]^{2}}\)
Question 7.
For the system 3A + 2B ⇌ C, the expression for equilibrium constant is
a) \(\frac{[3 \mathrm{~A}][2 \mathrm{~B}]}{[\mathrm{C}]}\)
b) \(\frac{[\mathrm{C}]}{[3 \mathrm{~A}][2 \mathrm{~B}]}\)
c) \(\frac{[\mathrm{A}]^{3}[\mathrm{~B}]^{2}}{[\mathrm{C}]}\)
d) \(\frac{[\mathrm{C}]}{[\mathrm{A}]^{3}[\mathrm{~B}]^{2}}\)
Answer:
d) \(\frac{[\mathrm{C}]}{[\mathrm{A}]^{3}[\mathrm{~B}]^{2}}\)
Question 8.
For the reaction PCl3(g) + Cl2(g) ⇌ PCl5(g) at 250°C, the value of Kc is 26, then the value of Kp at the same temperature will be
a) 0.61
b) 0.57
c) 0.83
d) 0.46
Answer:
a) 0.61
Question 9.
The equilibrium constant for the reaction N2(g) + O2(g) ⇌ 2NO(g) at temperature T is 4 × 10-4. The value of Kc for the reaction NO(g) ⇌ 1/2 N2(g) + 1/2 O2(g) at the same temperature is ___________.
a) 4 × 10-4
b) 50
c) 2.5 × 102
d) 0.02
Answer:
b) 50
Question 10.
In which of the following equilibria, the value of Kp is less than Kc?
a) H2 + I2 ⇌ 2HI
b) N2 + 3 H2 ⇌ 2NH3
c) N2 + O2 ⇌ 2NO
d) CO + H2O ⇌ CO2 + H2
Answer:
b) N2 + 3 H2 ⇌ 2NH3
![]()
Question 11.
In the reversible reaction A + B ⇌ C + D. The concentration of each C and D at equilibrium was 0.8-mole lit, then the equilibrium constant will be
a) 6.4
b) 0.64
c) 1.6
d) 16.0
Answer:
d) 16.0
Question 12.
The Kc for H2(g) + I2(g) ⇌ 2 HI(g), is 64. If the volume of the container is reduced to one – half of its original volume, the value of the equilibrium constant will be
a) +28
b) 64
c) 32
d) 16
Answer:
b) 64
Question 13.
For the reaction C(s) + CO2(g) ⇌ 2CO (g), the partial pressure of CO2 and CO are 2.0 and 4.0 atm respectively at equilibrium. The Kp for the reaction is
a) 0.5
b) 4.0
c) 8.0
d) 32.0
Answer:
c) 8.0
Question 14.
When the rate of forward reaction becomes equal to backward reaction, this state is termed as
a) chemical equilibrium
b) Reversible state
c) Equilibrium
d) All of these
Answer:
d) All of these
Question 15.
The equilibrium constant of the reaction H2(g) + I2(g) ⇌ 2 HI(g) is 64. If the volume of the container is reduced to one – fourth of its original volume, the value of the equilibrium constant will be
a) 16
b) 32
c) 64
d) 128
Answer:
c) 64
![]()
Question 16.
For the reaction PCl3(g) + Cl2(g) ⇌ PCl5(g), the position of equilibrium can be
shifted to the right by _______.
a) increasing the temperature
b) Doubling the volume
c) Addition of Cl2 at constant volume
d) Addition of equimolar quantities of PCl3 and PCl5
Answer:
b) Doubling the volume
Question 17.
For the reaction 2 NOCl(g) ⇌ 2 NO(g) + Cl2(g), Kc at 427 is 3 × 10-6 L mol-1. The value of Kp is nearly
a) 7.50 × 10-5
b) 2.50 × 10-5
c) 2.50 × 10-4
d) 1.72 × 10-4
Answer:
d) 1.72 × 10-4
Question 18.
Rate of reaction curve for equilibrium can be like [rf = forward rate, rb = backward rate]
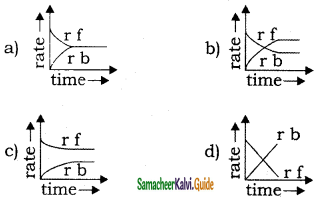
Answer:
a) 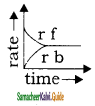
Question 19.
For a reaction, N2 + 3H2 ⇌ 2NH3(g), the value of Kc does not depend upon
A) initial concentration of the reactants
B) pressure
C) temperature
D) Catalyst
a) only C
b) A, B, C
c) A, B, C
d) A, B, C, D
Answer:
c) A, B, C
Question 20.
2NO2 ⇌ 2NO + O2, K = 1.6 × 10-12 NO + \(\frac{1}{2}\)O2 ⇌ NO2; K’ = ?
a) K’ = \(\frac{1}{\mathrm{~K}^{2}}\)
b) K’ = \(\frac{1}{\mathrm{~K}}\)
c) K’ = \(\frac{1}{\sqrt{\mathrm{K}}}\)
d) None of these
Answer:
c) K’ = \(\frac{1}{\sqrt{\mathrm{K}}}\)
![]()
Question 21.
At 1000 K, the value of Kp for the reaction, A(g) + 2B(g) ⇌ 3C (g) + D(g) is 0.05 atm. The value of Kc in terms of R would be
a) 20000 R
b) 0.02 R
c) 5 × 10-5 R
d) 5 × 10-5 × R-1
Answer:
d) 5 × 10-5 × R-1
Question 22.
In a chemical equilibrium, the rate constant for the backward reaction is 7.5 x 10-4 and the equilibrium constant is 1.5. The rate constant for the forward reaction
a) 2 × 10-3
b) 5 × 10-4
c) 1.12 × 10-3
d) 9.0 × 10-4
Answer:
c) 1.12 × 10-3
Question 23.
A reversible reaction is one which
a) Proceeds in one direction
b) proceeds in both direction
c) proceeds spontaneously
d) All the statements are wrong
Answer:
b) proceeds in both direction
Question 24.
The equilibrium constant in a reversible reaction at a given temperature
a) depends on the initial concentration of the reactants
b) depends on the concentration of the products at equilibrium
c) does not depend on the initial concentrations
d) It is not characteristic of the reaction
Answer:
c) does not depend on the initial concentrations
Question 25.
For the system A(g) + 2B(g) ⇌C (g), the equilibrium concentrations are (A) 0.06 mole / lit (B) 0.12 mole / lit and (C) 0.216 mole / lit. The Keq for the reaction is
a) 250
b) 416
c) 4 × 10-3
d) 125
Answer:
a) 250
![]()
Question 26.
A chemical reaction is at equilibrium when
a) Reactants are completely transformed into products
b) The rates of forward and backward reactions are equal
c) Formation of products is minimized
d) Equal amounts of reactants and products are present
Answer:
b) The rates of forward and backward reactions are equal
Question 27.
Partial pressures of A, B, C and D on the basis of gaseous system A + 2B ⇌ C + 3D are A = 0.20, B = 0.10, C = 0.30 and D = 0.50 atm. The numerical value of equilibrium constant is _______.
a) 11.25
b) 18.75
c) 5
d) 3.75
Answer:
b) 18.75
Question 28.
Molar concentration of 96 g of O2 contained in a 2 L vessel is ______.
a) 16 mol / L
b) 1.5 mol / L
c) 4 mol / L
d) 24 mol / L
Answer:
b) 1.5 mol / L
Question 29.
According to law of mass action rate of a chemical reaction is proportional to
a) concentration of reactants
b) molar concentration of reactants
c) concentration of products
d) molar concentration of products
Answer:
b) molar concentration of reactants
Question 30.
The equilibrium constant of the reaction SO2(g) + \(\frac{1}{2}\) O2(g) ⇌ SO3 (g) is 4 × 10-3 atm-1/2. The equilibrium constant of the reaction 2SO3(g) ⇌ 2SO2(g) + O2(g) would be
a) 250 atm
b) 4 × 103 atm
c) 0.25 × 104 atm
d) 6.25 × 104 atm
Answer:
d) 6.25 × 104 atm
![]()
Question 31.
The rate constant for forward and backward reactions of the hydrolysis of ester are 1.1 × 10-2 and 1.5 × 10-3 per minute respectively. The equilibrium constant for the reaction is ___________.
a) 4.33
b) 5.33
c) 6.33
d) 7.33
Answer:
d) 7.33
Question 32.
The active mass of 64 g of HI is a two litre of flask would be
a) 2
b) 1
c) 5
d) 0.25
Answer:
d) 0.25
Question 33.
At a certain temperature, 2HI ⇌ H2 + I2. Only 50 % HI is dissociated at equilibrium. The equilibrium constant is
a) 0.25
b) 1.0
c) 3.0
d) 0.50
Answer:
a) 0.25
Question 34.
Unit of equilibrium constant for the reversible reaction, H2 + I2 ⇌ 2HI is ________.
a) mol-1 litre
b) mol-2 litre
c) mol litre-1
d) none of these
Answer:
d) none of these
Question 35.
For the reaction
CO(g) + \(\frac{1}{2}\)O2(g) ⇌ CO2(g), Kp/Kc is _______.
a) RT
b) (RT)-1
c) (RT)-1/2
d) (RT)1/2
Answer:
c) (RT)-1/2
![]()
Question 36.
The following equilibrium are given
N2 +3H2 ⇌ 2NH2 ………K1
N2 + O2 ⇌ 2N0 ………….K2
H2 + 1/2 O2 ⇌ H2O …….. K3
The equilibrium constant of the reaction.
2 NH3 + 5/2 O2 ⇌ 2NO + 3H2O, in terms of K1, K2 and K3 is _________.
a) \(\frac{\mathbf{K}_{1} \mathbf{K}_{2}}{\mathbf{K}_{3}}\)
b) \(\frac{\mathrm{K}_{1} \mathrm{~K}_{3}^{2}}{\mathrm{~K}_{2}}\)
c) \(\frac{\mathrm{K}_{2} \mathrm{~K}_{3}^{2}}{\mathrm{~K}_{1}}\)
d) K1 K2 K3
Answer:
c) \(\frac{\mathrm{K}_{2} \mathrm{~K}_{3}^{2}}{\mathrm{~K}_{1}}\)
Question 37.
For the reaction
2NO2(g) ⇌ 2NO(g) + O2(g) Kc = 1.8 × 10-6 at 184°C and R = 0.083 Jk-1 mol-1. When Kp and are compared at 184C, it
is found that
a) Kp > Kc
b) Kp < Kc
c) Kp – Kc
d) Kp × Kc( depends upon pressure of gases)
Answer:
a) Kp > Kc
Question 38.
The rate of forward reaction is two times that of reverse reaction at a given temperature and identical concentration. K equilibrium is
a) 2.5
b) 2.0
c) 0.5
d) 1.5
Answer:
b) 2.0
Question 39.
In a reaction A+B ⇌ C+D,the concentrations of A, B, C and D [in mole/lit] are 0.5, 0.8, 0.4 & 1.0 respectively. Th. equilibrium constant is
a) 0.1
b) 1.0
c) 10
d) ∞
Answer:
b) 1.0
Question 40.
For the following thre. reactions 1, 2 and 3 equilibrium constants are given:
1. CO(g) + H2O(g) ⇌ CO(g) + H2(g); K1
2.CH4(g) + H2O(g) ⇌ CO + 3H(g); K2
3. CH4(g) + 2H2O(g) ⇌ CO(g) + 4H2(g); K3
Which of the following relations is correct?
a) K1√K2 = K3
b) K2K3 = K1
c) K3 = K1K2
d) K3 = K23 K12
Answer:
c) K3 = K1K2
![]()
Question 41.
4 moles of A is mixed with 4 moles of B. At equilibrium for the reaction A + B ⇌ C + D. 2 moles of C and D is formed. The equilibrium constant for the reaction will be
a) \(\frac{1}{4}\)
b) \(\frac{1}{2}\)
c) 1
d) 4
Answer:
c) 1
Question 42.
In a reaction PCl5 ⇌ PCl3 + Cl2 degree of dissociation is 30%. If Initial moles of PCl5 is one then total moles at equilibrium is
a) 1.3
b) 0.7
c) 1.6
d) 1.0
Answer:
a) 1.3
Question 43.
When 3 moles of A and 1 mole of B are mixed in 1 lit vessel, the following reaction takes place A(g) + B(g) ⇌ 2C(g), 1.5 moles of ‘C’ are formed. The equilibrium constant for the reaction is
a) 0.12
b) 0.25
c) 0.50
d) 4.0
Answer:
d) 4.0
Question 44.
In which of the following, the reaction proceeds towards completion
a) K = 103
b) K = 10-2
c) K = 10
d) K = 1
Answer:
a) K = 103
Question 45.
Two moles of NH3 when put into a previously evacuated vessel (1L) partially dissociate into N2 and H2. If at equilibrium one mole of NH3 is present, the equilibrium constant is ________.
a) \(\frac{3}{4}\) mol2 lit-2
b) \(\frac{27}{64}\) mol2 lit-2
c) \(\frac{27}{32}\) mol2 lit-2
d) \(\frac{27}{16}\) mol2 lit-2
Answer:
d) \(\frac{27}{16}\) mol2 lit-2
![]()
Question 46.
For the reaction N2(g) + O2(g) ⇌ 2NO(g) the value of Kc at 800°C is 0.1. When the equilibrium concentration of both the reactions is 0.5 mol, what is the value of Kp at the same temperature?
a) 0.5
b) 0.1
c) 0.01
d) 0.025
Answer:
b) 0.1
Question 47.
28 g of N2(g) and 6g of H2(g) were mixed at equilibrium 17 g NH3 was produced. The weight of N2 and H2 at equilibrium are respectively.
a) 11g, 0g
b) 1g, 3g
c) 14g, 3g
d) 11g, 3g
Answer:
c) 14g, 3g
Question 48.
2SO3 ⇌ 2SO2 + O2 If Kc = 100, α = 1, half of the reaction is completed, the concentration of SO3 and SO2 are equal, the concentration of O2 is _________.
a) 0.001 M
b) \(\frac{1}{2}\) SO2
c) 2 times of SO2
d) Data in complete
Answer:
d) Data in complete
Question 49.
For reaction HI ⇌ \(\frac{1}{2}\)H2 + \(\frac{1}{2}\)I2 value of Kc is \(\frac{1}{8}\), then value of K is \(\frac{1}{8}\) for H2 + I2 ⇌ 2HI.
a) \(\frac{1}{64}\)
b) 64
c) \(\frac{1}{8}\)
d) 8
Answer:
b) 64
Question 50.
2 moles of PCl5 were heated in a closed vessel of 2 liter capacity. At equilibrium, 40% of PCl5 is dissociated into PCl3 and Cl2. The value of equilibrium constant is
a) 0.266
b) 0.53
c) 2.66
d) 5.3
Answer:
a) 0.266
![]()
Question 51.
For a reaction H2 + I2 ⇌ 2HI at 721 k, the value of equilibrium constant is 50. If 0.5 mol each of H2 and I2 is added to the system the value of equilibrium constant will be
a) 40
b) 60
c) 50
d) 30
Answer:
c) 50
Question 52.
In a reaction the rate of reaction is proportional to its active mass, this statement is known as
a) Law of mass action
b) Le – Chatelier principle
c) Faradays’s law of electrolysis
d) law of constant proportion
Answer:
b) Le – Chatelier principle
Question 53.
In a chemical equilibrium A + B ⇌ C + D, when one mole each of the two reactants are mixed, 0.6 mole each of the products are formed. The equilibrium constant calculated is
a) 1
b) 0.36
c) 2.25
d) \(\frac{4}{9}\)
Answer:
c) 2.25
Question 54.
Under a given set of experimental conditions, with an increase in the concentration of the reactants, the rate of a chemical reaction.
a) Decreases
b) Increases
c) Remains unaltered
d) First decreases and then increases
Answer:
c) Remains unaltered
Question 55.
A + B ⇌ C + D, If finally the concentrations of A and B are both equal but at equilibrium concentration of D will be twice of that of A then what will be the equilibrium constant of reaction.
a) \(\frac{4}{9}\)
b) \(\frac{9}{4}\)
c) \(\frac{1}{9}\)
d) 4
Answer:
d) 4
![]()
Question 56.
Theory of ‘active mass’ indicates that the rate of chemical reaction is directly proportional to the ___________.
a) Equilibrium constant
b) Properties of reactants
c) Volume of apparatus
d) Concentration of reactants
Answer:
d) Concentration of reactants
Question 57.
In the reaction, A + B ⇌ 2C, at equilibrium the concentration of A and B is 0.20 mol L-1 each and that of C was found to be 0.60 mol L-1. The equilibrium constant of the reaction is
a) 2.4
b) 18
c) 4.8
d) 9
Answer:
d) 9
Question 58.
The rate at which substances react depends on their _______.
a) Atomic weight
b) Molecular weight
c) Equivalent weight
d) Active mass
Answer:
c) Equivalent weight
Question 59.
If in the reaction N2O4 ⇌ 2NO2, α is that part of N2O4 which dissociates, then the number of moles at equilibrium will be
a) 3
b) 1
c) (1- α)2
d) (1 + α)
Answer:
d) (1 + α)
Question 60.
On decomposition of NH4HS, the following equilibrium is established NH4HS(s) ⇌ NH3(g) + H2S(g). If the total pressure is P atm, then the equilibrium constant Kp is equal to _______.
a) P atm
b) P2 atm2
c) \(\frac{\mathrm{P}^{2}}{4}\) atm2
d) 2P atm
Answer:
c) \(\frac{\mathrm{P}^{2}}{4}\) atm2
![]()
Question 61.
Some gaseous equilibrium are given below:
i) CO + H2O ⇌ CO2 + H2
ii) 2CO + O2 ⇌ 2CO
iii) 2H2 + O2 ⇌ 2H3O
find out the relation between equilibrium constants.
a) K = K1K2
b) K = (K1K2)2
c) K = (K1K2)–\(\frac{1}{2}\)
d) K = \(\left(\frac{\mathrm{K}_{1}}{\mathrm{~K}_{2}}\right)^{\frac{1}{2}}\)
Answer:
d) K = \(\left(\frac{\mathrm{K}_{1}}{\mathrm{~K}_{2}}\right)^{\frac{1}{2}}\)
Question 62.
Kc = 9 for the reaction A + B ⇌ C + D. If A and B are taken In equal amounts, then amount of ‘C’ in equilibrium is
a) 1
b) 0.25
c) 0.75
d) none of these
Answer:
c) 0.75
Question 63.
In the reaction C(S) + CO2(g) ⇌ 2CO(g) the equilibrium pressure is 12 atm. If 50 % of CO2 reacts, then Kp will be ________.
a) 12 atm
b) 16 atm
c) 20 atm
d) 24 atm
Answer:
b) 16 atm
Question 64.
For the following gases equilibrium, N2O4(g) ⇌ 2NO2(g) Kp is found to be equal to Kc. This is attained when,
a) 0°C
b) 273 k
c) 1 k
d) 12.19 k
Answer:
d) 12.19 k
Question 65.
Consider the following reversible gaseous reactions {At 298 K)
a) N2O4 ⇌ 2NO2
b) 2SO2 + O2 ⇌ 2SO3
c) 2HI ⇌ H2 + I2
d) X + Y ⇌ 4Z
Answer:
a) N2O4 ⇌ 2NO2
![]()
Question 66.
For the reaction A + 2B ⇌ 2C at equilibrium [C] = 1.4 M, [A]0 = 1 M, [B]0 = 2M, [C]0 = 3 M. The value of Kc is .
a) 0.084
b) 8.4
c) 84
d) 840
Answer:
a) 0.084
Question 67.
For the reaction H2(g) + I2(g) ⇌ 2HI(g). Kc = 66.9 at 350°C and Kc = 50.0 at 448°C. The reaction has
a) ∆H = +ve
b) ∆H = -ve
c) ∆H = zero
d) ∆H = not found the signs.
Answer:
b) ∆H = -ve
Question 68.
In an equilibrium reaction H2(g) + I2(g) ⇌ 2HI(g), ∆H = -3000 calories, which factor favours dissociation of HI
a) Low temperature
b) High pressure
c) High temperature
d) Low pressure
Answer:
c) High temperature
Question 69.
In an equilibrium reaction for which ∆G°= 0, the equilibrium constant K Is
a) 0
b) 1
c) 2
d) 10
Answer:
b) 1
Question 70.
Consider the following reversible reaction at equilibrium, 2H2O(g) ⇌ 2H2(g) + O2(g). Which one of the following ∆H = 241. 7 kJ changes in a conditions will lead to maximum decomposition of H2O(g)?
a) Increasing both temperature and pressure
b) Decreasing temperature and increasing pressure
c) Increasing temperature and decreasing pressure
d) Increasing temperature at constant pressure.
Answer:
c) Increasing temperature and decreasing pressure
![]()
Question 71.
In which of the following system doubling the volume of the container causes a shift to the right.
a) H2(g) + Cl2(g) ⇌ 2HCl(g)
b) 2CO(g) + O2(g) ⇌ 2CO2(g)
c) N2(g) + 3H2(g) ⇌ 2NH3(g)
d) PCl5(g) ⇌ PCl3(g) + Cl2(g)
Answer:
d) PCl5(g) ⇌ PCl3(g) + Cl2(g)
Question 72.
∆G° (HI, g) ≈ 1.7 kJ. What is the equilibrium constant at 25° C for 2HI(g) ⇌ H2(g) + I2(g)
a) 24.0
b) 3.9
c) 2.0
d) 0.5
Answer:
d) 0.5
Question 73.
In the reaction A2(g) + 4B2(g) ⇌ 2AB4 ∆H < 0 the formation of AB4 is will, be favoured at
a) Low temperature, high pressure
b) High temperature, low pressure
c) Low temperature, low pressure
d) High temperature, high pressure
Answer:
a) Low temperature, high pressure
Question 74.
N2 + 3H2 ⇌ 2NH3. If temperature of following equilibrium reaction increases then the reaction.
a) Shifts Right side
b) Shifts left side
C) Remains unchanged
d) No change
Answer:
b) Shifts left side
Question 75.
Consider the equilibrium N2(g) + 3H2(g) ⇌ 2NH3 ∆H = -93.6 kJ. The maximum yield of ammonia is obtained by
a) Decrease of temperature and increase of pressure.
b) Increase of temperature and decreases of pressure.
c) Decrease of both the temperature and pressure
d) Increase of both the temperature and pressure
Answer:
a) Decrease of temperature and increase of pressure.
![]()
Question 76.
H2(g) + I2(g) ⇌ 2HI(g) ∆H = +q cal, then formation of HI
a) Is favoured by lowering the temperature
b) Is favoured by increasing the pressure
c) Is unaffected by change in pressure
d) Is unaffected by change in temperature
Answer:
c) Is unaffected by change in pressure
Question 77.
The formation of SO3 takes place according to the following reaction, 2SO2 + O2 ⇌ 2SO3; ∆H = 45.2 k cal. The formation of SO3 is favoured by
a) Increasing in temperature
b) Removal of oxygen
c) Increase of volume
d) Increase of pressure
Answer:
d) Increase of pressure
Question 78.
Which of the following equilibrium is not shifted by an increase in the pressure?
a) H2(g) + 3H2(g) ⇌ 2HI(g)
b) N2(g) + 3H2(g) ⇌ 2NH3(g)
c) 2CO(g) + O2(g) ⇌ 2CO2(g)
d) 2C(g) + O2(g) ⇌ 2CO(g)
Answer:
a) H2(g) + 3H2(g) ⇌ 2HI(g)
Question 79.
Consider the heterogeneous equilibrium in a closed container. NH4HS(s) ⇌ NH3(g) + H2S(g) if more NH4HS is added to the equilibrium
a) Partial pressure of NH3 increases
b) Partial pressure of H2S increases
c) Total pressure in the container increases
d) No effect on partial pressure of NH3 and H2S
Answer:
d) No effect on partial pressure of NH3 and H2S
Question 80.
According to Le – Chatelier’s principle adding heat to a solid and liquid in equilibrium with endothermic nature will cause the ____________.
a) Temperature to rise
b) Temperature to fall
c) Amount of solid to decrease
d) Amount of liquid to decrease
Answer:
c) Amount of solid to decrease
![]()
Question 81.
According to Le-chatelier’s principle, adding heat to a solid to liquid in equilibrium will causes the
a) temperature to increase
b) temperature to decrease
c) amount of liquid to increase
d) amount of solid to increase
Answer:
c) amount of liquid to increase
Question 82.
For the reaction A + B + Q ⇌ C + D, if the temperature is increased, then concentration of the products will
a) increase
b) decrease
c) Remain same
d) become zero
Answer:
a) increase
Question 83.
H2(g) + I2(g) ⇌ 2HI(g). In this reaction when pressure increases, the reaction direction
a) does not change
b) is forward
c) is backward
d) decreases
Answer:
a) does not change
Question 84.
Which of the following factor is shifted the reaction PCl3 + Cl2 ⇌ PCl5 at the left side?
a) Adding PCl5
b) increase pressure
c) constant temperature
d) catalyst
Answer:
a) Adding PCl5
Question 85.
The rate of reaction of which of the following is not affected by pressure?
a) PCl3 + Cl2 ⇌ PCl5
b) N2 + 3 H2 ⇌ 2NH3
c) N2 + O2 ⇌ 2NO
d) 2 SO2 + O2 ⇌ 2 SO3
Answer:
c) N2 + O2 ⇌ 2NO
![]()
Question 86.
Which reaction is not affected by change in pressure?
a) H2 + I2 ⇌ 2HI
b) 2 C + O2 ⇌ 2CO
c) N2 + 3 H2 ⇌ 2NH3
d) PCl5 ⇌ PCl3 + Cl2
Answer:
a) H2 + I2 ⇌ 2HI
Question 87.
Which of the following reactions proceeds at low pressure?
a) N2 + 3H2 ⇌ 2NH3
b) H2 + I2 ⇌ 2HI
c) PCl5 ⇌ PCl3 + Cl2
d) N2 + O2 ⇌ 2NO
Answer:
c) PCl5 ⇌ PCl3 + Cl2
Question 88.
In the following reaction PCl5 ⇌ PCl3(g) + Cl2(g) at constant temperature, rate of backward reaction, is increased by
a) inert gas mixed at constant volume
b) Cl2 gas mixed at constant volume
c) inert gas mixed at constant pressure
d) PCl5 mixed in constant volume
Answer:
b) Cl2 gas mixed at constant volume
Question 89.
On cooling of following system at equilibrium CO2(s) ⇌ CO2(g)
a) There is no effect on the equilibrium state
b) more gas is formed
c) more gas solidifies
d) None
Answer:
c) more gas solidifies
Question 90.
On the velocity in a reversible reaction the correct explanation of the effect of catalyst is _______.
a) it provides a new reaction path of low activation energy
b It increases a kinetic energy of the reacting molecules
c) it displaces the equilibrium state on right side
d) it decreases the velocity of backward reaction
Answer:
a) it provides a new reaction path of low activation energy
![]()
Question 91.
According to Le – Chatelier’s principle, if heat is given to solid – liquid system, then
a) Quantity of solid will reduce
b) Quantity of liquid will reduce
c) increase in temperature
d) decrease in temperature
Answer:
a) Quantity of solid will reduce
Question 92.
In a gives system, water and ice are in equilibrium, if pressure is applied to the system then .
a) more of ice is formed
b) amount of ice and water will remain same
c) more of ice is melted
d) either (a) or (c)
Answer:
c) more of ice is melted
Question 93.
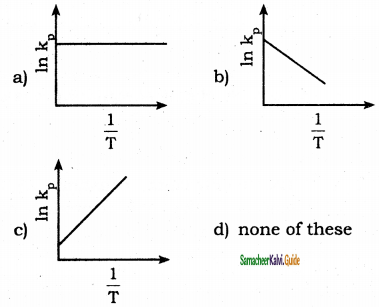
Answer:
b) 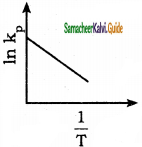
Question 94.
The value of ∆G° for a reaction in aqueous phase having Kc = 1, would be
a) – RT
b) -1
c) 0
d) + RT
Answer:
c) 0
Question 95.
The equilibrium constants for the reaction, Br2 ⇌ 2 Br at 500 K and 700 K are 1 × 10-10 and 1 × 10-5 respectively. The reaction is
a) endothermic
b) exothermic
c) fast
d) slow
Answer:
a) endothermic
![]()
Question 96.
In the reaction A2(g) + 4 B2(g) ⇌ 2AB4(g), ∆H < 0, the decomposition of AB4(g) will be favoured at
a) low temperature and high pressure
b) high temperature and low pressure
c) low temperature and low pressure
d) high temperature and high pressure
Answer:
c) low temperature and low pressure
Question 97.
The equilibrium constant for the reaction N2(g) + O2(g) ⇌ 2NO (g) is 4 × 10-4 at 200 K. In the presence of a catalyst the equilibrium is attained 10 times faster. Therefore the equilibrium constant in pressure of the catalyst at 200 K is
a) 4 × 10-3
b) 4 × 10-4
c) 4 × 10-5
d) None
Answer:
b) 4 × 10-4
Question 98.
Change in volume of the system does not alter the number of moles in which of the following equilibrium.
a) N2(g) + O2(g) ⇌ 2 NO (g)
b) PCl5(g) ⇌ PCl3(g) + Cl2(g)
c) N2(g) + 3H2(g) ⇌ 2 NH3(g)
d) SO2Cl2(g) ⇌ SO2(g) + Cl2(g)
Answer:
a) N2(g) + O2(g) ⇌ 2NO2 (g)
Question 99.
In which of the following equilibrium reactions the equilibrium would shift to the right, if total pressure is increased
a) N2 + 3H2 ⇌ 2 NH3
b) H2 + I2 ⇌ 2HI
c) H2 + Cl2 ⇌ 2 HCl
d) N2O4 ⇌ 2NO
Answer:
a) N2 + 3H2 ⇌ 2 NH3
Question 100.
The graph in K (eq) vs \(\frac{1}{T}\) relates for a reaction must be
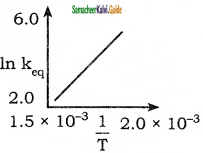
a) Exothermic
b) Endothermic
c) ∆H is negligible
d) High spontaneous at ordinary temperature
Answer:
a) Exothermic
Question 101.
Which of the following statements is cor¬rect for a reversible process in state of equilibrium?
a) ∆G° = -2.30 RT log K
b) ∆G° = 2.30 RT log K
c) ∆G = -2.30 RT log K
d) ∆G = 2.30 RT log K
Answer:
a) ∆G° = -2.30 RT log K
Question 102.
Which of the following is a characteristic of a reversible reaction?
a) Number of moles of reactants and products are equal
b) it can be influenced by a catalyst
c) it can never proceed to completion
d) none of the above
Answer:
c) it can never proceed to completion
Question 103.
The equilibrium constant in a reversible reaction at a given temperature
a) depends on the initial concentration of the reactants
b) depends on the concentration of the products at equilibrium
c) does not depend on the initial concentration
d) it is not characteristic of the reaction
Answer:
c) does not depend on the initial concentration
![]()
II. Very short question and answers(2 Marks):
Question 1.
What are reversible reactions?
Answer:
Reactions in which the products can react to give back reactants under suitable conditions are called reversible reactions.
Question 2.
What are irreversible reactions?
Answer:
Reactions in which the products formed do not react to give back reactants under normal conditions are called irreversible reactions.
Question 3.
Why the chemical equilibrium is referred to as Dynamic Equilibrium?
Answer:
At equilibrium the forward and the backward reactions are proceeding at the same rate and no macroscopic change is observed. So chemical equilibrium is often called as dynamic equilibrium.
Question 4.
What is equilibrium constant?
Answer:
The equilibrium constant Kc may be defined as the ratio of product of the active masses of the products to that of the reactants.
Question 5.
Write the Applications of equilibrium constant?
Answer:
The knowledge of equilibrium constant helps us in
- predicting the extent of a reaction, predicting the direction in which the net reaction is taking place and
- calculation of equilibrium constants and concentration of the reactants and products.
![]()
Question 6.
What is Le – Chatelier’s principle?
Answer:
It states that “If a system at equilibrium is subjected to a change of concentration, pressure or temperature, then the equilibrium is shifted in such a ways to nullify the effect of that disturbance.
Question 7.
For the H2(g) + I2(g) ⇌ 2HI(g) reaction the value of Kc at 717 K is 48. If at a particular instant, the concentration of H2, I2, and HI is 0.2 mol lit-1 and 0.6 mol lit-1 respectively, then calculate Q and predict the direction of reaction.
Solution:
Q = \(\frac{\left[\mathrm{H}_{1}\right]^{2}}{\left[\mathrm{H}_{2}\right]\left[\mathrm{I}_{2}\right]}=\frac{0.6 \times 0.6}{0.2 \times 0.2}\) = 9
Q < Kc, the reaction will proceed in the forward direction.
Question 8.
Define Degree of dissociation.
Answer:
The fraction of the total number of moles of the reactants are dissociated.
Degree of dissociation ‘x’ =
‘x’ = \(\frac{\text { no.of moles dissociated }}{\text { Total no. of moles present initially }}\)
Question 9.
Dissociation of PCl5 decreases in presence of increase in Cl2, Why?
Answer:
- The dissociation equilibrium of PCl5 in gaseous state is written as PCl5(g) ⇌ PCl3(g) + Cl2(g)
- In a chemical equilibrium increasing the concentration of the products results in shifting the equilibrium in favour of the reactants. So in the presence of increased Cl2 reversed reaction only favoured.
- Hence dissociation of PCl5 decreases in presence of increase in Cl2.
Question 10.
The equilibrium constant for Kc for A(g) ⇌ B(g) is 2.5 × 10-2 . The rate constant of the forward reaction is 0.05 sec-1. Calculate the rate constant of the reverse reaction.
Answer:
Kc = 2.5 × 10-2;
Kf = 0.05 sec-1
Kc = \(\frac{\mathrm{K}_{\mathrm{f}}}{\mathrm{K}_{\mathrm{r}}}\)
= \(\frac{0.05}{2.5 \times 10^{-2}}\) = 2
Rate constant of the reverse reaction,
Kr = 2 sec-1.
![]()
Question 11.
The equilibrium constant for the reaction 2SO3(g) ⇌ 2SO2(g) + O2(g) is 0.15 mol dm-3 at 900K. Calculate the equilibrium constant for the reaction 2SO2(g) + O2(g) ⇌ 2SO3(g) at the same temperature. 2SO3(g) ⇌ 2SO2(g) + O2(g).
Answer:
Kc = \(\frac{\left[\mathrm{SO}_{2}\right]^{2}\left[\mathrm{O}_{2}\right]}{\left[\mathrm{SO}_{3}\right]^{2}}\) = 0.15
K’ = \(\frac{1}{\mathrm{~K}_{\mathrm{c}}}=\frac{1}{0.15}\)
= 6.6 mole dm-3
K’ = 6.6 mole dm-3
III. Short question and answers(3 Marks):
Question 1.
What is solid – liquid equilibrium? Give example.
Answer:
- The equilibrium between solid and liquid at its melting point (or freezing point) at normal atmospheric pressure is called solid-liquid equilibrium.
- If some ice-cubes along with water are placed in a thermos flask (at 00 C and 1 atm), then there will be no change in the mass of ice and water.
- At equilibrium,
Rate of melting of ice = Rate of freezing of water
H2O(s) ⇌ H2O(l)
Question 2.
What is liquid – vapour equilibrium?
Answer:
The equilibrium of a liquid with its gas at its boiling point at 1 atm pressure is called liquid – vapour equilibrium.
Rate of evaporation = Rate of condensation
H2O(l) ⇌ H2O(g).
Question 3.
What is Solid – vapour equilibrium?
Answer:
- When solid iodine is placed in a closed transparent vessel, after sometime the vessel gets filled up with violet vapour.
- The intensity of the colour increases and finally becomes constant, i.e., equilibrium is attained.
I2(s) ⇌ I2(g)
Question 4.
What are Homogeneous and Heterogeneous equilibriums?
Answer:
Homogeneous equilibrium:
In a homogeneous equilibrium, all the reactants and products are in the same phase.
Example:
H2(g) + I2(g) ⇌ 2HI (g)
In the above equilibrium, H2, I2, and HI are in the gaseous state.
Similarly, for the following reactions,
CH3COOCH3(aq) + H2O(aq) ⇌ CH3COOH (aq) + CH3OH (aq) all the reactants and products are in homogenous solution phase.
Heterogeneous equilibrium:
If the reactants and products are present in different phases, it is known as heterogeneous equilibrium.
Example:
H2O(l) ⇌ H2O (g)
CaCO3(s) ⇌ CaO(s) + CO2(g).
Question 5.
What is Law of mass action?
Answer:
“At any instant, the rate of a chemical reaction at a given temperature is directly proportional to the product of the active masses of the reactants”.
Rate ∝ [Reactants]x
[ ] = \(\frac{\mathrm{n}}{\mathrm{v}}\), {Unit is mol.dm-3 or mol.L-1}
where,
n is the number of moles and
V is the volume of the container (dm3 or L).
![]()
Question 6.
What is the reaction quotient? Write the relation of K & Q.
Answer:
Reaction quotient ‘Q’ is defined as the ratio of the product of active mass of reaction products raised to the respective stoichiometric coefficients in the balanced chemical equation to that of the reactants under non equilibrium.
Reaction quotient Q.
Q = \(\frac{[\mathrm{C}]^{1}[\mathrm{D}]^{\mathrm{m}}}{[\mathrm{A}]^{\mathrm{x}}[\mathrm{B}]^{\mathrm{y}}}\)
If Q = Kc, the reaction is in equilibrium
If Q > Kc, the reaction will proceed in the reverse direction i.e., formation of reactants.
If Q < Kc, the reaction will proceed in the forward direction i.e., formation of products.
Question 7.
Explain effect of pressure of formation of HI.
Answer:
Pressure has no effect on the equilibrium when the total number of the moles of the gaseous reactants and the gaseous products are equal. ( ∆ng = 0).
Therefore, pressure has effect only on the equilibrium with ∆ng = 0.
Let us consider the following reactions
H2(g) + I2(g) ⇌ 2HI(g)
N2(g) + O2(g) ⇌ 2NO(g)
Here,
The number of moles on reactants and products are equal. So pressure has no effect on them.
Question 8.
The equilibrium concentrations of NH3, N2 and H2 are 1.8 × 10-2 M, 1.2 × 10-2 M and 3 × 10-2 M respectively. Calculate the equilibrium constant for the formation of NH3 from N2 and H2. [Hint: M= mol lit-1]
Solution:
Given data:
[NH3] = 1.8 × 10-2 M ;
[N2] = 1.2 × 10-2 M ;
[H2] = 3 × 10-2 M ;
Kc = ?
N2(g) + 3H2(g) ⇌ 2NH3(g)
Kc = \(\frac{\left[\mathrm{NH}_{3}\right]^{2}}{\left[\mathrm{~N}_{2}\right]\left[\mathrm{H}_{2}\right]^{3}}\)
Kc = \(\frac{1.8 \times 10^{-2} \times 1.8 \times 10^{-2}}{1.2 \times 10^{-2} \times 3 \times 10^{-2} \times 3 \times 10^{-2} \times 3 \times 10^{-2}}\)
Kc = 1 × 10-2 l2 mol-2
Question 9.
One mole of H2 and one mole of I2 are allowed to attain equilibrium. If the equilibrium mixture contains 0.4 mole of HI. Calculate the equilibrium constant.
Answer:
Given data:
[H2] = 1 mole;
[I2] = 1 mole
At equilibrium, [HI] = 0.4 mole; Kc = ?
H2(g) + I2 ⇌ 2HI(g)
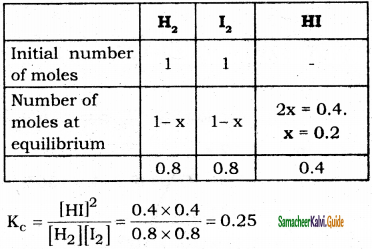
Question 10.
Relate the three equilibrium constants.
i) N2 + O2 ⇌ 2NO; K1
ii) 2NO + O2 ⇌ 2NO2; K2
iii) N2 + 2O2 ⇌ 2NO2; K3
Answer:
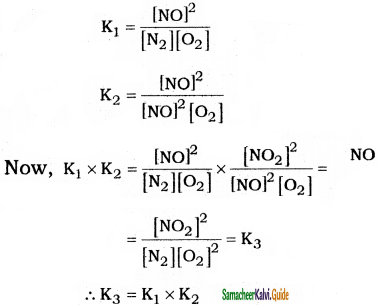
Question 11.
Write the equilibrium constant for the following:
i)H2O2(g) ⇌ H2O + 1/2O2(g)
ii) CO(g) + H2O(g) ⇌ CO2(g) + H2(g)
iii) N2O4(g) ⇌ 2NO2(g)
Answer:
i)H2O2(g) ⇌ H2O + 1/2O2(g)
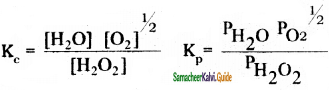
ii) CO(g) + H2O(g) ⇌ CO2(g) + H2(g)

iii) N2O4(g) ⇌ 2NO2(g)

Question 12.
Calculate the ∆ng, for the following reactions:
i) H2(g) + I2(g) ⇌ 2HI
ii) 2H2O(g) + 2Cl2(g) ⇌ 4HCl(g) + O2(g)
∆ng = (Total number of moles of gaseous products) – (Total number of moles of gaseous reactants)
Answer:
i) H2(g) + I2(g) ⇌ 2HI
∆ng = 2 – 2 = 0
∆ng = 0
ii) 2H2O(g) + 2Cl2(g) ⇌ 4HCl(g) + O2(g)
∆ng = 5 – 4 = 1
∆ng = 1
![]()
Question 13.
For the reaction A+B ⇌ 3C at 25°C, a 3 litre volume reaction vessel contains 1, 2, and 4 moles of A, B, and C respectively at equilibrium, calculate the equilibrium constant Kc of the reaction at 25°C.
Answer:
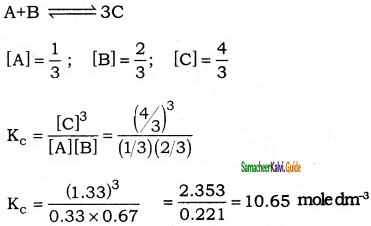
Question 14.
At temperature T1 the equilibrium constant of reaction is K1 At a higher temperature T2, K2 is 10% of K1. Predict whether the equilibrium is endothermic or exothermic.
Answer:
At temperature, T1, the equilibrium constant is K1
At a high temperature, T2, the equilibrium constant K2 = 10%KX
K2 = \(\frac{10}{100}\) K1
K2 = 0.1 K1
K1 > K2
Hence, the equilibrium is exothermic.
Question 15.
Calculate the Kc when a mixture containing 8.07 moles of H2 and 9.08 moles of I2 are reacted at 448°C until 13.38 moles of HI was formed at the equilibrIum.
Answer:
The equilibrium reaction is,
H2(g) + I2(g) ⇌ 2HI(g)
Kc = \(\frac{(2 x)^{2}}{(a-x)(b-x)}\)
a = 8.07 moles;
b = 9.08 moles
2x = 13.38 moles
x = 6.69 moles
Kc = \(\frac{4(6.69)^{2}}{(8.07-6.69)(9.08-6.69)}\)
= \(\frac{4 \times 44.75}{1.38 \times 2.39}\)
Kc = 54.29
Question 16.
For the equilibrium 2NOCl(g) ⇌ 2NO(g) + Cl2(g) the value of the equilibrium constant Kc is 3.75 × 10-6 at 790°C. Calculate Kp for this equilibrium at the same temperature. [Hint: Kp = Kc(RT)∆ng]
Answer:
Kc = 3.75 × 10-6;
T = 790 + 273 = 1063 K
Kp = Kc × (RT)∆ng
For the reaction,
2NOCl(g) ⇌ 2NO(g) + Cl2(g)
∆ng = 3 – 2 = 1
Kp = 3.75 × 10-6 × (8.314 × 1063)
= 33141.6 × 10-6
= 3.31 × 104 × 10-6
Kp = 3.31 × 10-2
Question 17.
How much PCl5 must be added to one litre volume reaction vessel at 250°C in order to obtain a concentration of 0.1 mole of Cl2, Kc for PCl5 ⇌ PCl3 + Cl2 is 0.0414 mol dm-3 at 250°C.
Answer:
PCl5 ⇌ PCl3 + Cl2
Number of moles of PCl5 at equilibrium = a – 0.1
Number of moles PCl3 at equilibrium = 0.1
Number of moles Cl2 at equilibrium = 0.1
Kc = \(\frac{\left[\mathrm{PCl}_{3}\right]\left[\mathrm{Cl}_{2}\right]}{\left[\mathrm{PCl}_{5}\right]}\)
= 0.0414
= \(\frac{0.1 \times 0.1}{a-0.1}\)
0.01 = (a – 0.1) 0.0414
0.01 = 0.0414a – 0.00414
0.0414a = 0.01414
a = 0.3415 mole
Question 18.
At 540 K, the equlllbrium constant Kp for PCl5 dissociation equilibrium at 1.0 atm is 1.77 atm. Calculate equilibrium constant in molar concentration (Kc) at same temperature and pressure.
Answer:
Kp = 1.77 atm;
T = 540 K;
a = 0.082
PCl5 ⇌ PCl3 + Cl2
∆ng = 2 – 1 = 1
Kp = Kc(RT)∆ng
1.77 = Kc × (0.0821 × 450)1
Kc = \(\frac{1.77}{(0.0821 \times 540)}\)
= 4 × 10-2 moles/litres.
![]()
III. Long question and answers (5 Marks):
Question 1.
Derive the Equilibrium constants Kp and K for the homogeneous reactions.
Answer:
Let us consider a reversible reaction,
xA + yB ⇌ lC + mD
where A and B are the reactants, C and D are the products, and x, y, l, and m are the coefficients of A, B, C, and D respectively.
Applying Law of mass action, the rate of the forward reaction,
rf α [A]x [B]y (or)
rf = kf [A]x [B]y
Similarly, the rate of the backward reaction,
rb a [C]l [D]m (or)
rb = k, [C]l [D]m
where kf and kb are proportionality constants
At equilibrium, Rate of forward reaction(rf) = Rate of backward reaction(rb)
kf α [A]x [B]y = kb a [C]l [D]m (or)
\(\frac{\mathrm{k}_{\mathrm{f}}}{\mathrm{k}_{\mathrm{b}}}=\frac{[\mathrm{C}]^{l}[\mathrm{D}]^{m}}{[\mathrm{~A}]^{x}[\mathrm{~B}]^{y}}\) = Kc
where, Kc is the equilibrium constant in terms of concentration.
At a given temperature, the ratio of the product of active masses of products to that the reactants is a constant, known as equilibrium constant. Each concentration term is raised to the power which is equal to the respective stoichiometric coefficients in the balanced chemical equation. Later when we study chemical kinetics we will learn that this is only approximately true. If the reactants and products behave as gases, then the equilibrium constant in terms of partial pressures can be written as
\(\frac{\mathrm{P}_{\mathrm{C}}^{l} \times \mathrm{P}_{\mathrm{D}}^{m}}{\mathrm{P}_{\mathrm{A}}^{x} \times \mathrm{P}_{\mathrm{B}}^{y}}\)where, Pc is the partial pressure of the gas, C and so on.
Question 2.
Derive Kp & Kc relation for the formation of HI.
Answer:
Synthesis of HI:
Consider that ‘a’ moles and ‘b’ moles are the initial concentration of H2 and I2 respectively, taken in a container of volume V. Let ‘x’ moles of each of H2 and I2 react together to form 2x moles of HI.
H2(g) + I2(g) ⇌ 2HI(g)

The equilibrium constant Kp can also be calculated as follows:
Total number of moles at equilibrium = a – x + b – x + 2x = a + b.
If the total pressure of the system at equilibrium be P, then
Partial pressure of H2,
pH2 = \(\left(\frac{a-x}{a+b}\right)\)P
Partial pressure of I2,
pI2 = \(\left(\frac{b-x}{a+b}\right)\)P
Partial pressure of HI,
pHI = \(\left(\frac{2x}{a+b}\right)\)P
Kp = \(\frac{\left(\frac{2 x}{a+b}\right)^{2} \mathrm{P}^{2}}{\left(\frac{a-x}{a+b}\right) \mathrm{P}\left(\frac{b-x}{a+b}\right) \mathrm{P}}=\frac{4 x^{2}}{(a-x)(b-x)}\)
In the above equilibrium, Kc and Kp are thus shown to be equal.
![]()
Question 3.
Derive the equilibrium constant Kp & Kc for the Dissociation of PCl5.
Answer:
Dissociation of PCl5:
Consider that ‘a’ moles is the initial concentration of PCl5 taken in a container of volume V. Let ‘x’ moles of PCl5 be dissociated into x moles each of PCl3 and Cl2.
PCl5(g) ⇌ PCl3(g) + PCl2(g)

The equilibrium constant Kp can also be calculated as follows:
Total number of moles at equilibrium = a – x + x + x = a + x
If the total pressure of the system at equilibrium be P, then
Partial pressure of PCl5,
pPCl5 = \(\left(\frac{a-x}{a+x}\right)\)P;
Partial pressure of PCl3,
pPCl3 = \(\left(\frac{x}{a+x}\right)\)P;
Partial pressure of Cl2
pCl2 = \(\left(\frac{x}{a+x}\right)\)P
Kp = \(\frac{\left(\frac{x}{a+x}\right) \mathrm{P}\left(\frac{x}{a+x}\right) \mathrm{P}}{\left(\frac{a-x}{a+x}\right) \mathrm{P}}=\frac{x^{2} \mathrm{P}}{(a-x)(a+x)}\)
Question 4.
Explain the effect of Concentration of formation of HI?
Answer:
Effect of concentration:
At equilibrium, the concentration of the reactants and the products does not change. The addition of more reactants or products to the reacting system at equilibrium causes an increase in their respective concentrations.
According to Le Chatelier’s principle, the effect of increase in concentration of a substance is to shift the equilibrium in a direction that consumes the added substance. Suppose at equilibrium if one of the reactants is added, then the equilibrium will shift in the forward direction and if one of the products is added, then the equilibrium will shift in backward direction.
Let us consider the reaction,
H2(g) + I2(g) ⇌ 2HI(g)

On addition of H2 or I2 to the equilibrium mixture product HI will tend to increase. In order to minimize the stress, the system shifts the reaction in a direction where H2 and I2 are consumed i.e., the formation of HI would relieve the stress. Hence, the equilibrium shifts to the right (forward direction).
Similarly, removal of HI (product) also boosts the forward reaction and increases the concentration of products.
If HI is added to the equilibrium mixture, the concentration HI is increased, then the system proceeds in the reverse direction to nullify the stress.
Question 5.
The equilibrium constant at 298 K for a reaction is 100. A + B ⇌ C + D. If the initial concentration of all the four species were 1 M each what will be equilibrium concentration of D (in mol lit-1)
Solution:
Given data:
[A] = [B] = [C] = 1 M;
Kc = 100; [D] = ?

10(1 – x) = 1 + x
10 – 10x – 1 – x = 0
9 – 11x = 0
11x = 9
x = 9/11 = 0.818
So, [D] at equilibrium = 1 + 0.818 = 1.818 M
![]()
Question 6.
What happens when ∆ng = 0, ∆ng = -ve, ∆ng= +ve In a gaseous reaction?
Answer:
(i) When ∆ng = 0, the total number of moles of gaseous products are equal to the total number of moles of gaseous reactants.
Eg:
H2(g) + I2(g) ⇌ 2HI(g)
∆ng = 2 – 2 = 0
Kp = Kc(RT)∆ng
If ∆ng = 0,
Kp = Kc(RT)0
∴ Kp = Kc
(ii) When ∆ng = +ve, the total number of moles of gaseous products are greater than the total number of moles of gaseous reactants.
Eg:
2H2O + 2Cl2(g) ⇌ 4HCl(g) + O2(g)
∆ng = 5 – 4 = 1
Kp = Kc(RT)1
Kp = Kc(RT)
∴ Kp > Kc
(iii) When ∆ng = -ve, the total number of moles of gaseous products are greater than the total number of moles of gaseous reactants.
N2(g) + 3H2(g) ⇌ 2NH3(g)
∆ng = 2 – 4 = -2
Kp = Kc(RT)-2
Kp = \(\frac{K_{c}}{(R T)^{2}}\)
∴ Kp < Kc
Question 7.
What is the relationship between the dissociation and formation equilibrium constant?
Answer:
In a chemical equilibrium reaction, the equilibrium constant for the dissociation equilibrium reaction which is also known as dissociation constant is found to be the reciprocal value of the equilibrium constant for the formation equilibrium reaction.
Formation of equilibrium reaction,
2SO2(g) + O2 ⇌ 2SO3
Kc = \(\frac{\left[\mathrm{SO}_{3}\right]^{2}}{\left[\mathrm{SO}_{2}\right]^{2}\left[\mathrm{O}_{2}\right]}\) dm3 /mole.
Dissociation equilibrium reaction of SO3 the reactants became products and vice versa.
2SO3(g) ⇌ 2SO2(g) + O2(g)
Kc‘ = \(\frac{\left[\mathrm{SO}_{2}\right]^{2}\left[\mathrm{O}_{2}\right]}{\left[\mathrm{SO}_{3}\right]^{2}}\) mole/dm3
= \(\frac{1}{\mathrm{~K}_{\mathrm{c}}}\)
Kc‘ is considered as the dissociation constant of SO3 gas. Usually, the equilibrium constant of the dissociation equilibrium is the reciprocal of the equilibrium constant of the formation equilibrium reaction.
Question 8.
In the equilibrium H2 + I2 ⇌ 2HI, the number of moles of H2, I2, and HI are 1, 2, 3 moles respectively. Total pressure of the reaction mixture is 60 atm. Calculate the partial pressures of H2, I2, and HI in the mixture.
Solution:
H2 + I2 ⇌ 2HI
Number of moles of H2 at equilibrium = 1 mole
Number of moles of I2 at equilibrium = 2 mole
Number of moles of HI at equilibrium = 3 mole
Total number of moles of equilibrium = 1 + 2 + 3 = 6 moles
Mole fraction of H2 = 1/6;
Mole fraction of I2 = 2/6;
Mole fraction of HI = 3/6
Partial pressure = Mole fraction × Total pressure
PH2 = \(\frac{1}{6}\) × 60 = 10 atm;
PI2 = \(\frac{2}{6}\) × 60 = 20 atm;
PHI = \(\frac{3}{6}\) × 60 = 30 atm.
![]()
Question 9.
In 1 litre volume reaction vessel, the equilibrium constant K of the reaction PCl5 ⇌ PCl3 + Cl2 is 2 × 10-4 What will be the degree of dissociation assuming only a small extent of 1 mole of PCl5 has dissociated?
Answer:
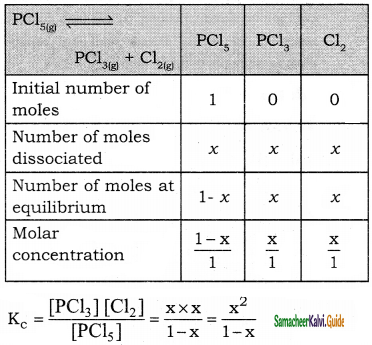
since ‘x’ is very small compared to 1,
then K2 = x2
2 × 10-4 = x2
Degree of dissociation x = \(\sqrt{2 \times 10^{-4}}\)
= 1.414 × 10-2
![]()
Question 10.
At 35°C, the value of K for the equilibrium reaction N2O4 ⇌ 2NO2 is 0.3174, Calculate the degree of dissociation when P is 0.2382 atm.
Answer:
| N2O4 ⇌ 2NO2 | N2O4 | NO2 |
| Initial number of moles | 1 | 0 |
| Number of moles dissociated | x | 2x |
| Number of moles equilibrium | 1 – x | 2x |
Total number of moles at equilibrium = 1 – x + 2x = 1 + x
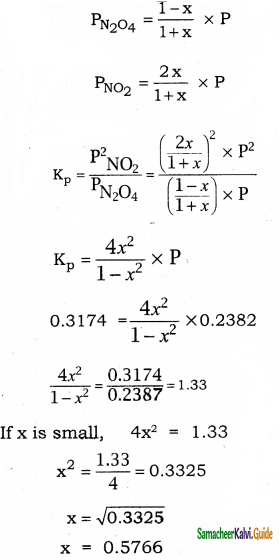
Question 11.
Degree of dissociation of PCl5 at 1 atm and 25°C is 0.2. Calculate the pressure at which PCl5 is half dissociated at 25°C.
Answer:
For PCl5 dissociation equilibrium,
Kp = \(\frac{x^{2} p}{1-x^{2}}\) [ P = total pressure = 1 atm]
x = 0.2
Kp = \(\frac{(0.2)^{2}(1.0)}{1-(0.2)^{2}}\)
= \(\frac{0.04}{1-0.04}\)
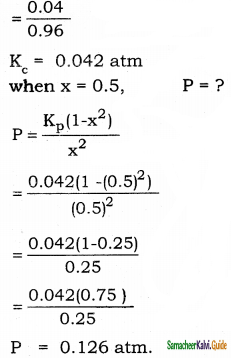
Question 12.
Initially, 0.1 moles each of H2 and I2 gases and 0.02 moles of HI gas are mixed in a reaction vessel of constant volume at 300K. Predict the direction towards which the reaction proceeds [K = 3.5 x 10-2].
Answer:
The formation of equilibrium is
H2(g) + I2(g) ⇌ 2HI(g)
Kc = \(\frac{[\mathrm{HI}]_{\mathrm{e}}^{2}}{\left[\mathrm{H}_{2}\right]_{\mathrm{e}}\left[\mathrm{I}_{2}\right]_{\mathrm{e}}}\)
= 3.5 × 10-2 at 300 K.
Under non equilibrium conditions;
Q = \(\frac{[\mathrm{HI}]^{2}}{\left[\mathrm{H}_{2}\right]\left[\mathrm{I}_{2}\right]}\)
= \(\frac{\left(2 \times 10^{-2}\right)^{2}}{0.1 \times 0.1}\)
= 4 × 10-2
Thus Q > Kc.
Therefore, the reaction will proceed initially before attaining the equilibrium in a direction such that the Q value is reduced. That is, concentrations of H2 and I2 should be increases, which is a reverse reaction. The Traction proceeds in the left side of fomiation equilibrium of HI and HI decomposes initially to H2 and I2 until Q = Kc.