Students get through the TN Board 11th Bio Botany Important Questions Chapter 8 Biomolecules which is useful for their exam preparation.
TN State Board 11th Bio Botany Important Questions Chapter 8 Biomolecules
Answer the following short answers.
Question 1.
What is meant by the cellular pool?
Answer:
The cell components are made of a collection of molecules called a cellular pool, which consists ’ of both inorganic and organic compounds.
Question 2.
What are Micronutrients? Give any two examples.
Answer:
Micronutrients, which are required in trace amounts, eg. Cobalt, zinc, boron, copper, molybdenum and manganese) and are essential for enzyme action.
Question 3.
Mention any four properties of water.
Answer:
- Adhesion and cohesion property.
- High latent heat of vaporisation.
- High melting and boiling point.
- Universal solvent.
![]()
Question 4.
Define primary metabolites.
Answer:
Primary metabolites are those that are required for the basic metabolic processes like photosynthesis, respiration, protein and lipid metabolism of living organisms.
Question 5.
Match the following:
| (i) Enzymes | (a) Abrin |
| (ii) Amino acid | (b) Morphine |
| (iii) Alkaloids | (c) Peroxidase |
| (iv) Toxins | (d) Leucine |
(a) (i)-(c), (ii)-(d), (iii)-(b), (iv)-(a)
(b) (i)-(d), (ii)-(c), (iii)-(b), (iv)-(a)
(c) (i)-(b), (ii)-(a), (iii)-(d), (iv)-(c)
(d) (i)-(c), (ii)-(a), (iii)-(d), (iv)-(b)
Answer:
Question 6.
Define monosaccharide.
Answer:
Monosaccharides are relatively small molecules constituting a single sugar unit. Glucose has a chemical formula of CgH^Og. It is a six-carbon molecule and hence is called hexose.
Question 7.
Define glycosidic bond.
Answer:
The bond formed between the glucose and fructose molecule by the removal of water is called a glycosidic bond. This is another example of a strong covalent bond.
Question 8.
What is chitin made up of?
Answer:
Chitin is a homo polysaccharide with amino acids added to form mucopolysaccharide. The ’ basic unit is a nitrogen-containing glucose derivative known as N-acetyl glucosamine. It forms the exoskeleton of insects and other arthropods. It is also present in the cell walls of fungi.
![]()
Question 9.
How does Herbivores digest cellulose?
Answer:
Herbivores can digest them With the help of bacteria present in the gut which produces en2yme cellulase. This is an example of mutualism.
Question 10.
What are steroids? Explain with an example.
Answer:
These are complex compounds commonly found in the cell membrane and animal hormones, eg. Cholesterol which reinforces the structure of the cell membrane in animal cells and in an unusual group of cell wall deficient bacteria —Mycoplasma.
Question 11.
Define the term amphoteric.
Answer:
(NH2), an acidic carboxylic group (COOH) and a hydrogen atom (H) and side-chain or variable R group* The amino acid is both an acid and a base is called amphoteric.
Question 12.
What do you know about the primary structure of a protein?
Answer:
The primary structure is the linear arrangement of amino acids in a polypeptide chain.
Question 13.
What is meant by an ionic bond?
Answer:
It is formed between any charged groups that are not joined together by a peptide bond. It is stronger than a hydrogen bond and can be broken by changes in pH and temperature.
![]()
Question 14.
Explain briefly anabolic reactions.
Answer:
Anabolic (building up of organic molecules). Synthesis of proteins from amino acids and synthesis of polysaccharides from simple sugars are examples of anabolic reactions.
Question 15.
What are the lock and key mechanism?
Answer:
The substrate binds to the specially formed pocket in the enzyme – the active site, this is called the lock and key mechanism of enzyme action. As the enzyme and substrate form an ES complex, the substrate is raised in energy to a transition state and then breaks down into products plus an unchanged enzyme.
Question 16.
What are competitive inhibitors? Give an example.
Answer:
Molecules that resemble the shape of the substrate and may compete to occupy the active site of the enzyme are known as competitive inhibitors, eg. the enzyme that catalyses the reaction between carbon dioxide and the CO2 acceptor molecule in photosynthesis, known as ribulose biphosphate carboxylase oxygenase (RUBISCO) is competitively inhibited by oxygen/carbon-di-oxide in the chloroplast.
Question 17.
Define the terms nucleotide and nucleoside?
Answer:
DNA and RNA are polymers of monomers called nucleotides, each of Which is composed of a nitrogen base, a pentose sugar and a phosphate. A purine or a pyrimidine and ribose or deoxyribose sugar are called nucleoside.
Question 18.
Mention any two sulphur-containing amino acids.
Answer:
Methionine and cysteine.
![]()
Question 19.
What is meant by Plectonemic coiling of DNA?
Answer:
Plectonemic coiling – the two strands of the DNA are wrapped around each other in a helix, making it impossible to simply move them apart without breaking the entire structure.
Question 20.
Give a short note on RNA.
Answer:
tRNA (transfer RNA): Translates the code from mRNA and transfers amino acids to the ribosome to build proteins. It is highly folded into an elaborate 3D structure and comprises about 15% of total RNA. It is also called soluble RNA.
Answer In brief.
Question 1.
Explain Polysaccharide with an example.
Answer:
- These are made of hundreds of monosaccharide units. Polysaccharides also called “Glycans”.
- Long-chain of branched or unbranched monosaccharides is held together by glycosidic bonds.
- Polysaccharide is an example of a giant molecule, a macromolecule and consists of only one type of monomer.
- Polysaccharides are insoluble in water and are sweet. Cellulose is an example built from repeated units of glucose monomer.
- Depending on the function, polysaccharides are of two types: Storage Polysaccharide and structural Polysaccharide.
![]()
Question 2.
Describe the test for reducing sugars.
Answer:
Aldoses and ketoses are reducing sugars. This means that, when heated with an alkaline solution of copper (II) sulphate (a blue solution called benedict’s solution), the aldehyde or ketone group reduces Cu2+ ions to Cu+ ions forming a brick-red precipitate of copper (I) oxide. In the process, the aldehyde or ketone group is oxidised to a carboxyl group (- COOH). This reaction is used as a test for reducing sugar and is known as Benedict’s test. The results of benedict’s test depend on the concentration of the sugar. If there is no reducing sugar it remains blue.
- Sucrose is not a reducing sugar.
- The greater the concentration of reducing sugar, the more is the precipitate formed and greater is the colour change.
Question 3.
Define triglycerides. Explain with examples.
Answer:
Triglycerides are composed of a single molecule of glycerol bound to 3 fatty acids. These include fats and oils. Fatty acids are long-chain hydrocarbons with a carboxyl group at one end which binds to one of the hydroxyl groups of glycerol, thus forming an ester bond. Fatty acids are structural unit of lipids and are carboxylic acid of long-chain hydrocarbons. The hydrocarbon can vary in length from 4-24 carbons and the fat may be saturated or unsaturated. In saturated fatty acids, the hydrocarbon chain is single-bonded.
eg. Palmitic acid, Stearic acid and unsaturated fatty acids.
eg. Oleic acid, linoleie acid) the hydrocarbon chain is double bonded (one/two/three). In general solid fats are saturated and oils are unsaturated, in which most are globules.
Question 4.
Enumerate the properties of enzymes.
Answer:
Properties of Enzyme:
- All are globular proteins.
- They act as catalysts and effective even in small quantity.
- They remain unchanged at the end of the reaction.
- They are highly specific.
- They have an active site where the reaction takes place.
- Enzymes lower the activation energy of the reaction they catalyse.
Question 5.
What are Allosteric enzymes? Explain with a suitable example.
Answer:
They modify enzyme activity by causing a reversible change w the structure of the enzyme active site. This in turn affects the ability of the substrate to bind to the enzyme. Such compounds are called Allosteric inhibitors, eg. The enzyme hexokinase which catalysis glucose to glucose-6 phosphate in glycolysis is inhibited by glucose 6 phosphate. This is an example of a feedback allosteric inhibitor.
![]()
Question 6.
Distinguish between nucleoside and nucleotide with two examples.
Answer:
| Nucleoside | Nucleotide |
| It is a combination of base and sugar. | It is a combination of nucleoside and phosphoric acid. |
| eg. Adenosine = Adenine + Ribose Guanosine = Guanine + Ribose | eg. Adenylic acid = Adenosine + phosphoric acid Guanylic acid = Guanosine + phosphoric acid |
Question 7.
Explain ribosomal RNA. Add a note on its function.
Answer:
RNA (ribosomal RNA): Single-stranded, metabolically stable, make up the two subunits of ribosomes. It constitutes 80% of the total RNA. It is a polymer with varied length from 120-3000 nucleotides and gives ribosomes their shape. Genes for rRNA are highly conserved and employed for phylogenetic studies.
Question 8.
Explain the process of negative feedback inhibition with a schematic diagram.
Answer:
Negative Feedback Inhibition: When the end product of a metabolic pathway begins to accumulate, it may act as an allosteric inhibitor of the enzyme controlling the first step of the pathway. Thus the product starts to switch off its own production as it builds up. The process is- self – regulatory. As the product is used up, its production is switched on once again. This is called end-product inhibition.
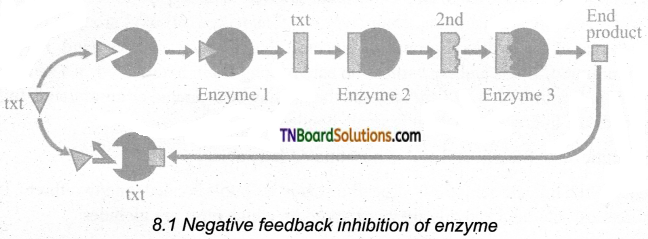
![]()
Question 9.
Explain the Michaelis-Menton Constant (km) with graphical representation.
Answer:
The rate of reaction is directly proportional to the enzyme concentration.
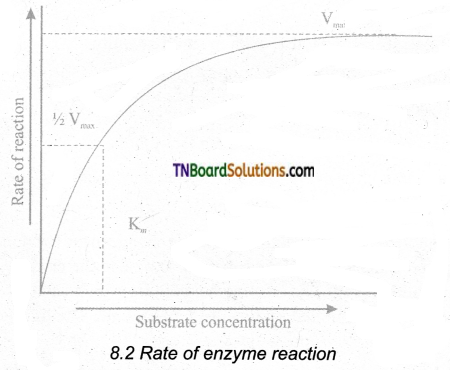
When the initial rate of reaction of an enzyme is measured over a range of substrate concentrations (with a fixed amount of enzyme) and the results plotted on a graph. With increasing substrate concentration, the velocity increases – rapidly at lower substrate concentration.
Question 10.
What is the three types of chemical bond in protein structure? Explain them with an example.
Answer:
A hydrogen bond is formed between some hydrogen atoms of oxygen and nitrogen in a polypeptide chain. The hydrogen atoms have a small positive charge and oxygen and nitrogen have a small negative charge. Opposite charges attract to form hydrogen bonds. Though these bonds are weak, a large number of them maintains the molecule in 3D shape.
Ionic Bond: It is formed between any charged groups that are not joined together by a peptide bond. It is stronger than a hydrogen bond and can be broken by changes in pH and temperature.
Disulfide Bond; Some amino acids like cysteine and methionine have sulphur. These form a disulphide bridge between sulphur atoms and amino acids.
Hydrophobic Bond: This bond helps some protein to maintain structure. When globular proteins are in solution, their hydrophobic groups point inwards away from water.
![]()
Answer In detail.
Question 1.
Describe the structure of a protein with a neat diagram.
Answer:
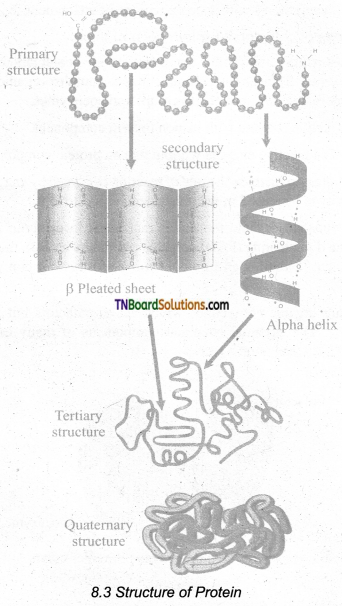
- Protein is synthesised on the ribosome as a linear sequence of amino acids which are held together by peptide bonds. After synthesis, the protein attains conformational change into a specific 3D form for proper functioning. According to the mode of folding, four levels of protein’ organisation have been recognised namely primary, secondary, tertiary and quaternary.
- The primary structure is the linear arrangement of amino acids in a polypeptide chain.
- Secondary structure arises when various functional groups are exposed on the outer surface of the molecular interaction by forming hydrogen bonds. This causes the amino acid chain to twist into a coiled configuration called a-helix or to fold into flat β-pleated sheets.
- Tertiary protein structure arises when the secondary level proteins fold into a globular structure called domains.
- Quaternary protein structure may be assumed by some complex proteins in which more than one polypeptide forms a large multi-unit protein. The individual polypeptide chains of the protein are called subunits and the active protein itself is called a multimer.
- eg. Enzymes serve as a catalyst for chemical reactions in the cell and are non-specific. Antibodies are complex glycoproteins with specific regions of attachment for various organisms.
Question 2.
What are the types of cofactors? Explain each of them.
Answer:
- Many enzymes require non-protein components called cofactors for their efficient activity. Cofactors may vary from simple inorganic ions to complex organic molecules. They are of three types: inorganic ions, prosthetic groups and coenzymes.
- Holoenzyme: Active enzyme with its non-protein component.
- Apoenzyme: The inactive enzyme without its non-protein component.
- Inorganic ions help to increase the rate of the reaction catalysed by enzymes. Example: Salivary amylase activity is increased in the presence of chloride ions.
- Prosthetic groups are organic molecules that assist in the catalytic function of an enzyme. Flavin adenine dinucleotide (FAD) contains riboflavin (Vit B2), the function of which is to accept hydrogen. ‘Haem’ is an iron-containing prosthetic group with an iron atom at its centre.
- Coenzymes are organic compounds that act as cofactors but do not remain attached to the enzyme. The essential chemical components of many coenzymes are vitamins, eg. NAD, NADP, Coenzyme A, ATP.
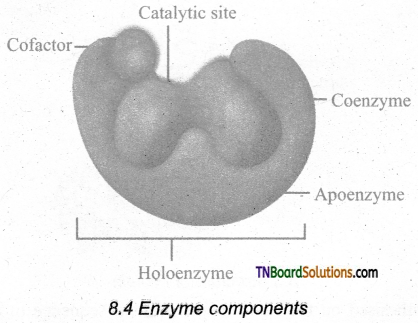
![]()
Question 3.
Explain the structure of DNA with Watson and Crick modal.
Answer:
- Watson and Crick shared the Nobel Prize in 1962 for their discovery, along with Maurice Wilkins, who had produced the crystallographic data supporting the model, Rosalind Franklin (1920-1958) had earlier produced the first clear crystallographic evidence of a helical structure. James Watson and Francis Crick of Cavendish laboratory in Cambridge built a scale model of the double-helical structure of DNA which is the most prevalent form of DNA, the B-DNA. This is the secondary structure of DNA.
- As proposed by James Watson and Francis Crick, DNA consists of a right-handed double helix with 2 helical polynucleotide chains that are coiled around a common axis to form a right-handed B form of DNA. The coils are held together by hydrogen bonds which occur between complementary pairs of nitrogenous bases. The sugar is called 2-deoxyribose because there is no hydroxyl at position 2′. Adenine and thiamine base pairs have two hydrogen bonds while guanine and cytosine base pairs have three hydrogen bonds.
Chargaff’s Rule:- A = T; G = C
- A+G = T + C
- A : T = G : C =1
- As published by Erwin Chargaff in 1949, a purine pairs with pyrimidine and vice versa. Adenine (A) always pairs with Thymine (T) by a double bond and Guanine (G) always pairs with Cytosine (C) by a triple bond.
Question 4.
Explain any two factors affecting the rate of enzyme reaction, with the help of graphical representation.
Answer:
Enzyme Reactions: Enzymes are sensitive to environmental condition. It could be affected by temperature, pH, substrate concentration and enzyme concentration. The rate of enzyme reaction is measured by the amount of substrate changed or amount of product formed, during a period of time.
Temperature: Heating increases molecular motion. Thus the molecules of the substrate and enzyme move more quickly resulting in a greater probability of occurrence of the reaction. The temperature that promotes maximum activity is referred to as optimum temperature.
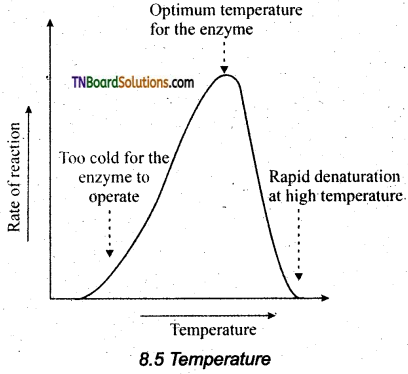
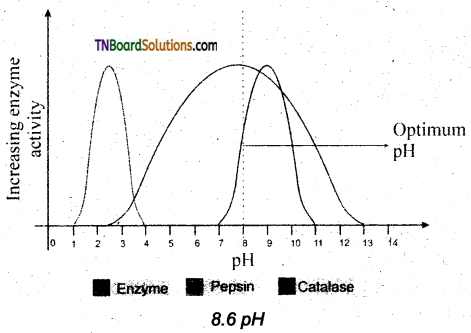
pH: The optimum pH is that at which the maximum rate of reaction occurs. Thus the pH change leads to an alteration of enzyme shape, including the active site. If extremes of pH are encountered by an enzyme, then it will be denatured.
Substrate Concentration: For a given enzyme concentration, the rate of an enzyme reaction increases with increasing substrate concentration.
Enzyme Concentration: The rate of reaction is directly proportional to the enzyme concentration:
![]()
Question 5.
Describe the structure and functions of various other polysaccharides.
Answer:
| Other Polysaccharides | Structure | Functions |
| Inulin | The polymer of fructose. | It is not metabolised in the human body and is readily filtered through the kidney. |
| Hyaluronic acid | Heteropolymer of d glucuronic acid and D-N acetyl glucosamine. | It accounts for the toughness and flexibility of cartilage and tendon. |
| Agar | Mucopolysaccharide from red algae. | Used as a solidifying agent in culture medium in the laboratory. |
| Heparin | Glucosamine glycan contains variably sulphated disaccharide unit present in the liver. | Used as an anticoagulant. |
| Chondroitin sulphate | Sulphated glycosaminoglycan composed of altering sugars (N-acetylglucosamine and glucuronic acid). | Dietary supplement for treatment of osteoarthritis. |
| Keratan sulphate | Sulphated glycosaminoglycan and is a structural carbohydrate | Acts as a cushion to absorb mechanical shock. |
Choose the correct answer.
1. Which is the most abundant component in living organisms.
(a) Minerals
(b) Macromolecules
(c) Water
(d) Protein
Answer:
(c) Water
2. In a water molecule, the hydrogen and oxygen atom stick together by:
(a) Monovalent bond
(b) Covalent bond
(c) Hydrogen bond
(d) None of the above
Answer:
(b) Covalent bond
3. Morphine is the first alkaloid to be found from a plant called:
(a) Vinca rosea
(b) Sweet pea
(c) Delonix regia
(d) Papaver somniferum
Answer:
(d) Papaver somniferum
![]()
4. Indicate a macromolecule:
(a) Amino acid
(b) Protein
(c) Nucleotide
(d) Glucose
Answer:
(b) Protein
5. The number of sugar units present in oligosaccharides:
(a) 14 to 15
(b) 6 to 8
(c) 2 to 10
(d) 11 to 12
Answer:
(c) 2 to 10
6. Sucrose is a
(a) Polysaccharide
(b) Disaccharide
(c) Monosaccharide
(d) Triglyceride
Answer:
(b) Disaccharide
7. A test for the presence of starch by adding a solution of iodine gives:
(a) Greenish blue colour
(b) Reddish green colour
(c) blue-black colour
(d) Violet-pink colour
Answer:
(c) blue-black colour
8. Glycogen is not seen in the organs of the human body:
(a) Muscle fibre
(b) Liver
(c) Brain
(d) Kidney
Answer:
(c) Brain
9. Chitin is composed of
(a) Mucopolysaccharides
(b) Oligopolysaceharides
(c) Glycoprotein
(d) Dipolysaccharides
Answer:
(a) Mucopolysaccharides
![]()
10. Match the following.
| (i) Inulin | (a) heteropolymer of D glucose |
| (ii) Hyaluronic acid | (b) Mucopolysaccharides |
| (Hi) Heparin | (c) Polymer of fructose |
| (iv) Agar | (d) Glycosamine glycon |
(a) (i)-(c), (ii)-(a), (iii)-(d), (iv)-(b)
(b) (i)-(d), (ii)-(c), (iii)~(b), (iv)-(a)
(c) (i)-(b), (ii)-(a), (iii)-(d), (iv)-(c)
(d) (i)-(c), (ii)-(a), (iii)-(d), (iv)-(b)
Answer:
(a) (i)-(c), (ii)-(a), (iii)-(d), (iv)-(b)
11. Lipids do not include:
(a) Steroid
(b) Waxes
(c) Enzymes
(d) phospholipids
Answer:
(c) Enzymes
12. A molecule of glycerol bound to have:
(a) 5 fatty acids
(b) 6 fatty acids
(c) 4 fatty acids
(d) 3 fatty acids
Answer:
(d) 3 fatty acids
13. Indicate saturated fatty acids:
(a) Palmitic acid
(b) Oleic acid
(c) Linoleic acid
(d) None of the above
Answer:
(a) Palmitic acid
![]()
14. Phospholipids serve as a major structural component of:
(a) Feathers
(b) Cell membrane
(c) Leaves
(d) Skin
Answer:
(b) Cell membrane
15. Cholesterol is an example of:
(a) Membrane lipids
(b) Triglycerides
(c) Steroids
(d) Adipose tissue
Answer:
(c) Steroids
16. The term ‘protein’ was coined by:
(a) Watson
(b) Gerardus Johannes Mulder
(c) Erwin Chargaff
(d) Maurice Wilkins
Answer:
(b) Gerardus Johannes Mulder
17. First protein insulin was sequenced by;
(a) Fred Sanger
(b) Robert Brown
(c) Robert Hooke
(d) Christian Anfinsen
Answer:
(a) Fred Sanger
18. Protein is synthesized in:
(a) Mitochondria
(b) Golgi body
(c) Lysosome
(d) Ribosome
Answer:
(d) Ribosome
19. A linear arrangement of amino acids is a polypeptide chain is seen in:
(a) Secondary structure of the protein
(b) Primary structure of the protein
(c) Tertiary structure of the protein
(d) Quaternary protein structure
Answer:
(b) Primary structure of the protein
![]()
20. Protein denaturation is due to:
(a) Exposure to pressure
(b) Exposure to light
(c) Exposure to heat
(d) None of the above
Answer:
(c) Exposure to heat
21. In a polypeptide chain hydrogen bone is formed between some hydrogen atoms of:
(a) Oxygen and methane
(b) Ethylene and nitrogen
(c) Nitrogen and methane
(d) Oxygen and nitrogen
Answer:
(d) Oxygen and nitrogen
22. Disulfide bond is seen between some amino acids like:
(a) Glycine and alanine
(b) Serine and proline
(c) Cysteine and methionine
(d) Aspartate and glutamate
Answer:
(c) Cysteine and methionine
23. Synthesis of polysaccharides from simple sugars is termed as:
(a) Catabolic reaction
(b) Anabolic reaction
(c) Hydrolytic reaction
(d) Oxidative reaction
Answer:
(b) Anabolic reaction
24. Indicate the correct statement:
(a) The rate of reaction is indirectly proportional to the enzyme concentration.
(b) The rate of reaction is directly proportional to the enzyme concentration.
(c) The rate of reaction is indirectly proportional to an increase in temperature
(d) None of the above.
Answer:
(c) The rate of reaction is indirectly proportional to an increase in temperature
![]()
25. The increased concentration of malonate inhibits the reaction of the enzyme, succinic dehydrogenase. This type of inhibitors is termed as:
(a) Competitive inhibitors
(b) Non-competitive inhibitors
(c) Irreversible inhibitors
(d) None of the above
Answer:
(a) Competitive inhibitors
26. NADP serves as:
(a) Apoenzyme
(b) Holoenzyme
(c) Coenzyme
(d) None of the above
Answer:
(c) Coenzyme
27. Formation of new chemical bonds using ATP as a source of energy is the mode of action of the enzymes.
(a) Hydrolase
(b) Isomerase
(c) Lyase
(d) Ligase
Answer:
(d) Ligase
28. DNA and RNA are polymers of monomers called:
(a) Nucleoside
(b) Nucleotide
(c) Pyrimidine
(d) Dinucleotide
Answer:
(b) Nucleotide
![]()
29. Which of the RNA constitutes 80% of the total RNA:
(a) mRNA
(b) tRNA
(c) rRNA
(d) None of the above
Answer:
(c) rRNA
30. Who got a noble prize for the finding of the helical structure of DNA?
(a) Rosalind Franklin and Erwin Chargaff
(b) Maurice Wilkins and Rosalind Franklin
(c) James Watson and Francis Crick
(d) Robert Hooke and Robert Brown
Answer:
(c) James Watson and Francis Crick